– For the study of cancer or ischemia –
- Easily available for live-cell imaging
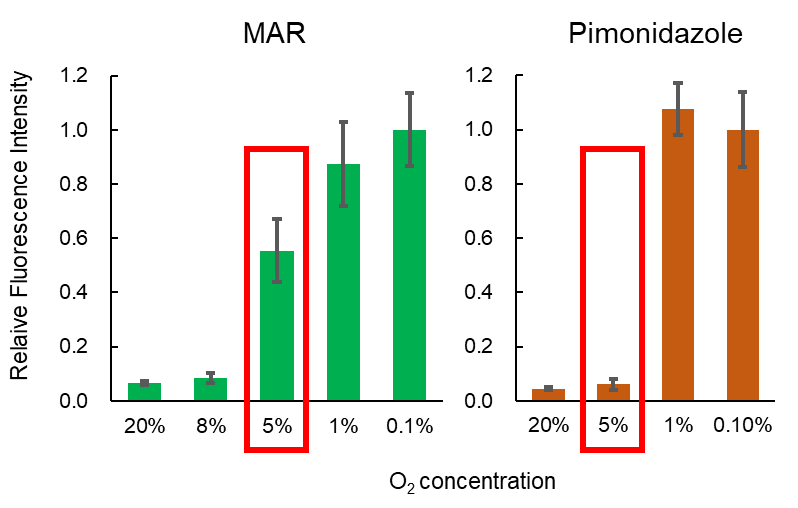
- As sensitive as pimonidazole
- Available for flow cytometry
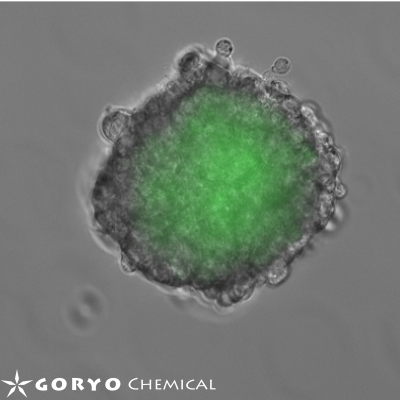
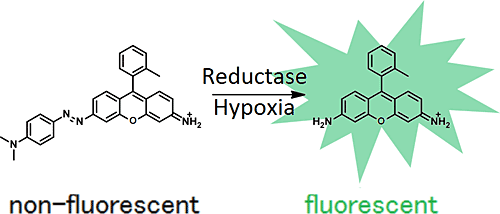
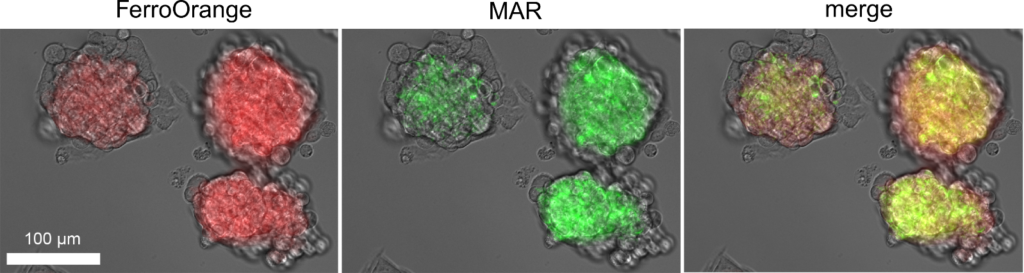
Hypoxia in living tissue closely relates to a wide array of pathologies, including cancer and ischemia. When using with the existing hypoxia detection probe, pimonidazole, immunostaining is necessary, and the probe is only available for the fixed cells. However, live-cell imaging of hypoxia is easily available with MAR.
Available through Merck KGaA (Darmstadt, Germany) as:
SCT033 BioTracker™ 520 Green Hypoxia Dye
![]()





 Contact Us
Contact Us
