H. Tanaka, S. Maeda, K. Nakamura, H. Hashizume, K. Ishikawa, M. Ito, K. Ohno, M. Mizuno, Y. Motooka, Y. Okazaki, S. Toyokuni, H. Kajiyama, F. Kikkawa, M. Hori (2021)
Plasma Process. Polym. in press DOI: 10.1002/ppap.202100056
Y. Ikeda, H. Hamano, Y. Horinouchi, L. Miyamoto, T. Hirayama, H. Nagasawa, T. Tamaki, K. Tsuchiya (2021)
J. Trace Elem. Med. Biol. 67: 126798 DOI: 10.1016/j.jtemb.2021.126798
F. Ito, K. Kato, I. Yanatori, T. Murohara, S. Toyokuni (2021)
Redox Biol. 47: 102174 DOI: 10.1016/j.redox.2021.102174
S. Zhuang, Y. Ma, Y. Zeng, C. Lu, F. Yang, N. Jiang, J. Ge, H. Ju, C. Zhong, J. Wang, J. Zhang S. Jiang (2021)
Cell Biol. Toxicol. in press DOI: 10.1007/s10565-021-09660-7.
A. Li, C. Liangy, L. Xu, Y. Wang, W. Liu, K. Zhang, J. Liu, J. Shi (2021)
Acta Pharm. Sin. B 11: 1329-1340 DOI: 10.1016/j.apsb.2021.03.017
K. Komoto, T. Nomoto, S. E. Muttaqien, H. Takemoto, M. Matsui, Y. Miura, N. Nishiyama (2021)
Cancer Sci. 112: 410–421 DOI: 10.1111/cas.14607
K. Tomita, T. Nagasawa, Y. Kuwahara, S. Torii, K. Igarashi, M. H. Roudkenar, A. M. Roushandeh, A. Kurimasa, T. Sato (2020)
Int. J. Mol. Sci. 22: 8300-8314 DOI: 10.3390/ijms22158300
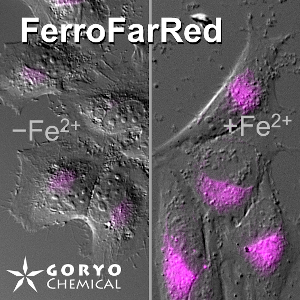
T. Hirayama, M. Niwa, S. Hirosawa, H. Nagasawa (2020)
ACS Sens. 5: 2950-2958 DOI: 10.1021/acssensors.0c01445
Y. Takashi, K.Tomita, Y. Kuwahara, M. H. Roudkenar, A. M. Roushandeh, K. Igarashi, T. Nagasawa, Y. Nishitani, T. Sato (2020)
Free Radic. Biol. Med. 161:60-70 DOI: 10.1016/j.freeradbiomed.2020.09.027
Y. Hirata, K. Kuwabara, M. Takashima, T. Murai (2020)
Chem. Res. Toxicol. 33: 2892–2902
Y. Hirata, Y. Ito, M. Takashima, K. Yagyu, K. Oh-Hashi, H. Suzuki, K. Ono, K. Furuta, M. Sawada (2020)
ACS Chem. Neurosci. 11: 76−85 DOI: 10.1021/acschemneuro.9b00619
K. Tomita, M. Fukumoto, K. Itoh. Y. Kuwahara, K. Igarashi, T. Nagasawa, M. Suzuki, A. Kurimasa, T. Sato (2019)
Biochem. Biophys. Res. Commun. 518: 712 DOI: 10.1016/j.bbrc.2019.08.117
K. F. Yambire, C. Rostosky, T. Watanabe, D. P. Grau, S. T. Odio, A. S. Guerrero, O. Senderovich, E. G. M. Holtz, I. Milosevic, J. Frahm, A. P. West, N. Raimundo (2019)
Elife 8: e51031 DOI: 10.7554/eLife.51031
M. Takashima, K. Ichihara , Y. Hirata (2019)
Food Chem. Toxicol. 132: 110669 DOI: 10.1016/j.fct.2019.110669
M. Sato, T. Hirayama, T. Fujii, H. Nagasawa, I. Minoura (Jan. 2018)
ASCB | EMBO 2017 Meeting (poster)
![]()





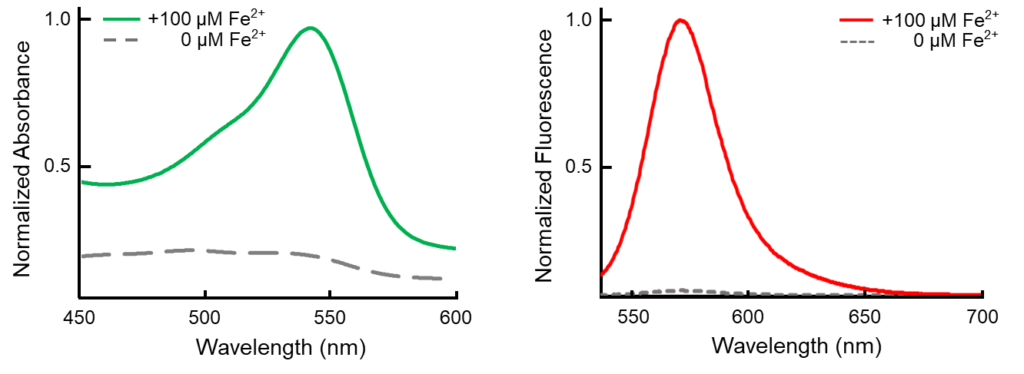
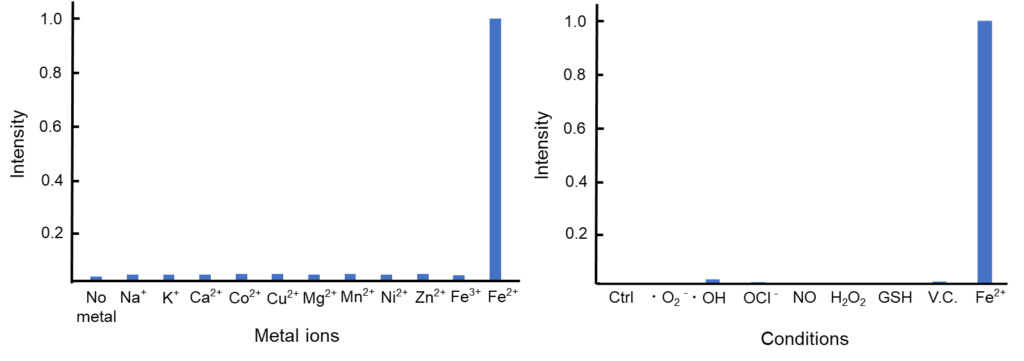
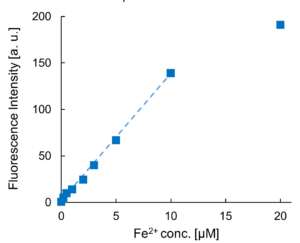
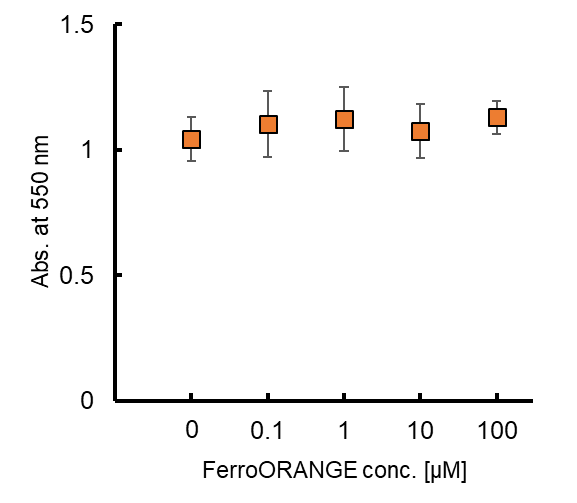
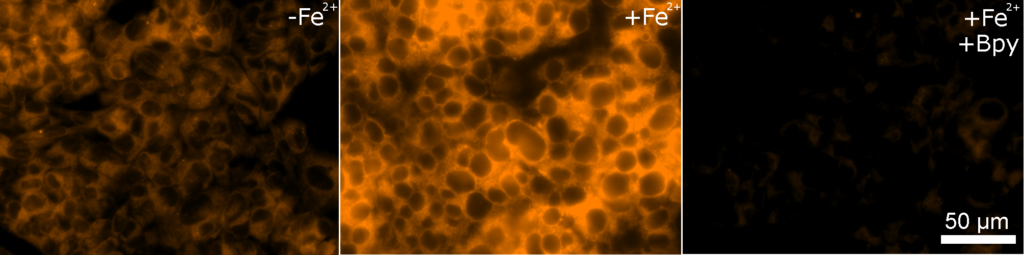
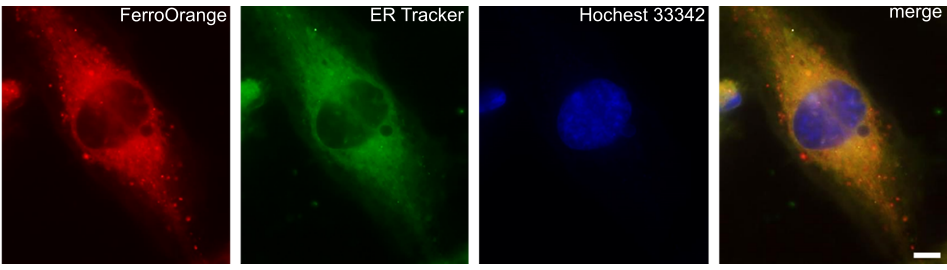
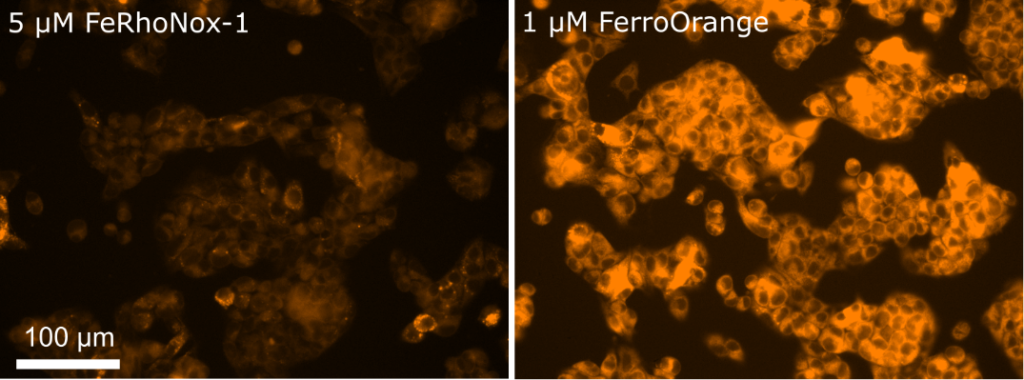
 Contact Us
Contact Us
