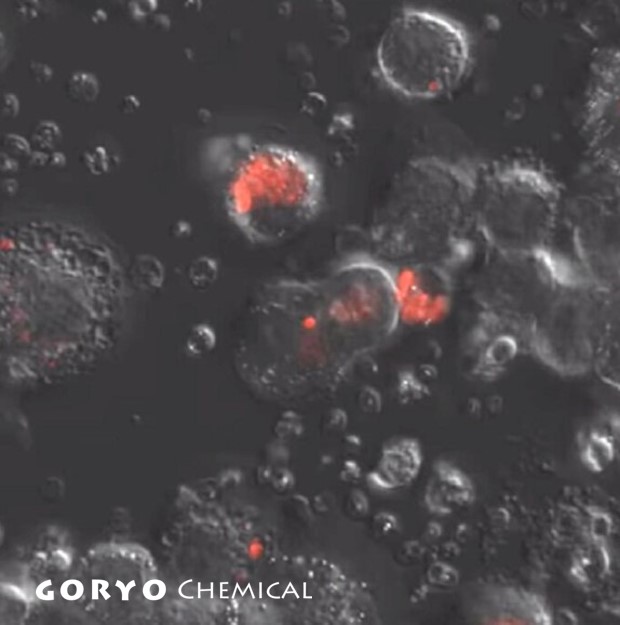
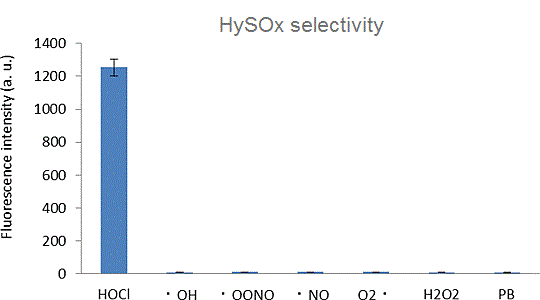
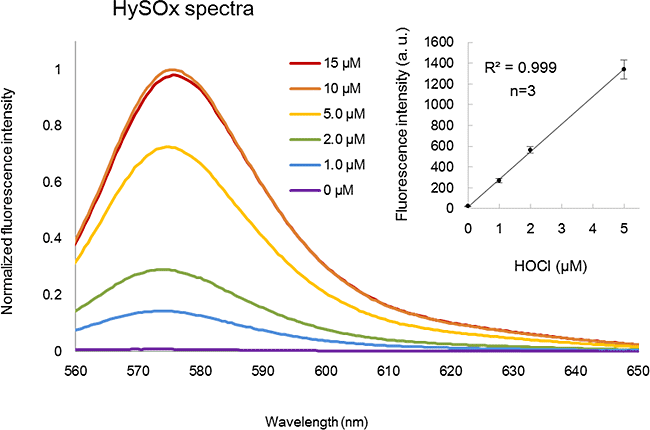
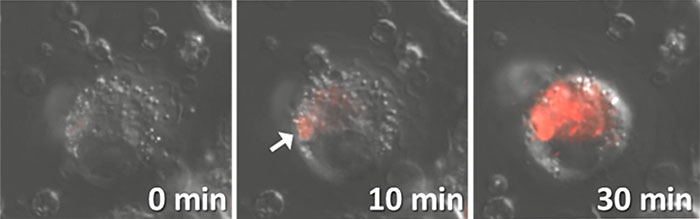
HySOx is a fluorescent probe that shows excellent selectivity against hypochlorous acid (HOCl). Hypochlorous acid is one of the various reactive oxygen species (ROS) including hydroxyradical (・OH), superoxide (O2-・), hydrogen peroxide (H2O2), singlet oxygen (1O2), nitric oxide (・NO), and peroxynitrite (ONOO-). HySOx is almost non-fluorescent in neutral solutions. When HySOx reacts with hypochlorous acid, it generates a highly fluorescent product (with excitation maximum: 553 nm and emission maximum: 574 nm).
Available through Merck KGaA (Darmstadt, Germany) as:
SCT034 BioTracker™ 574 Red HOCL Dye
![]()





 Contact Us
Contact Us
