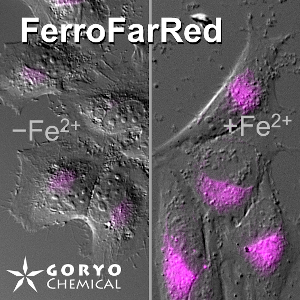
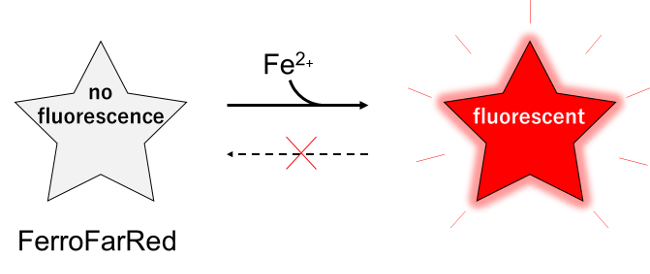
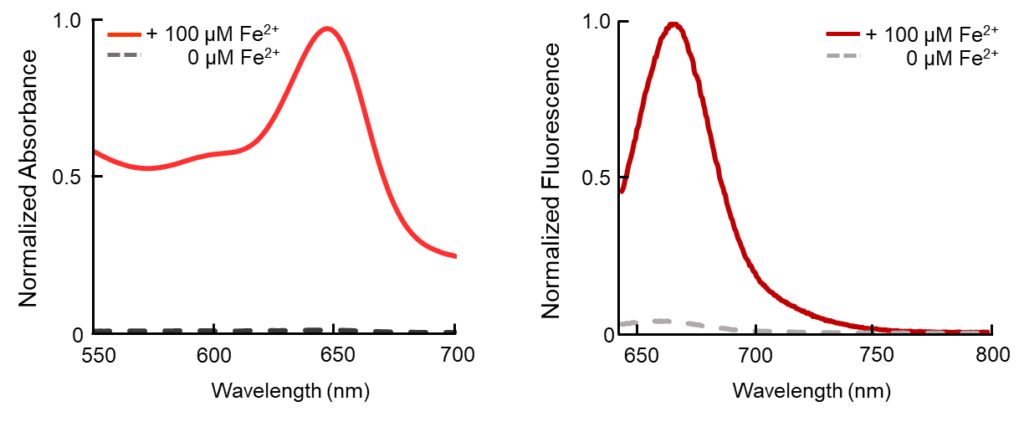
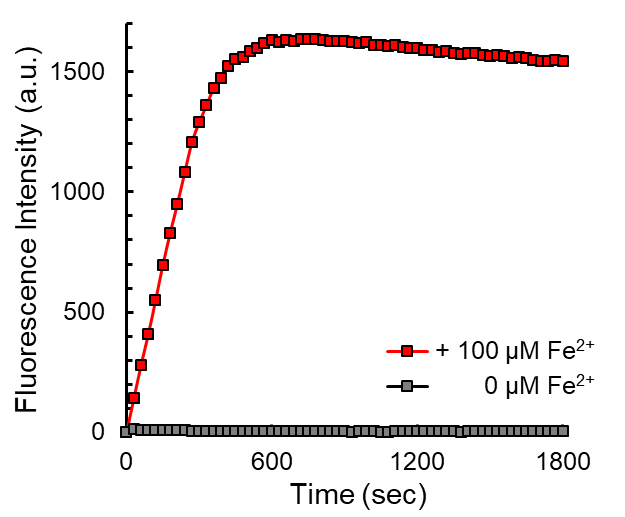
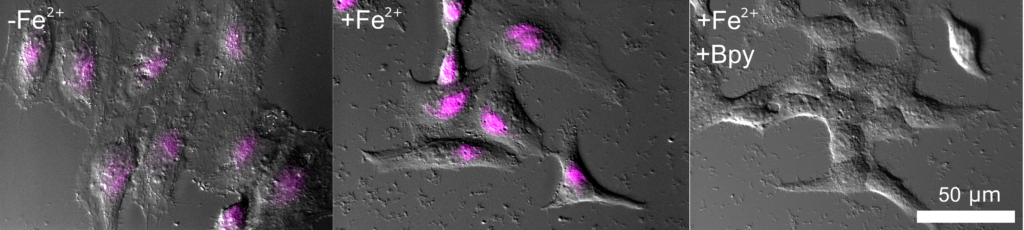
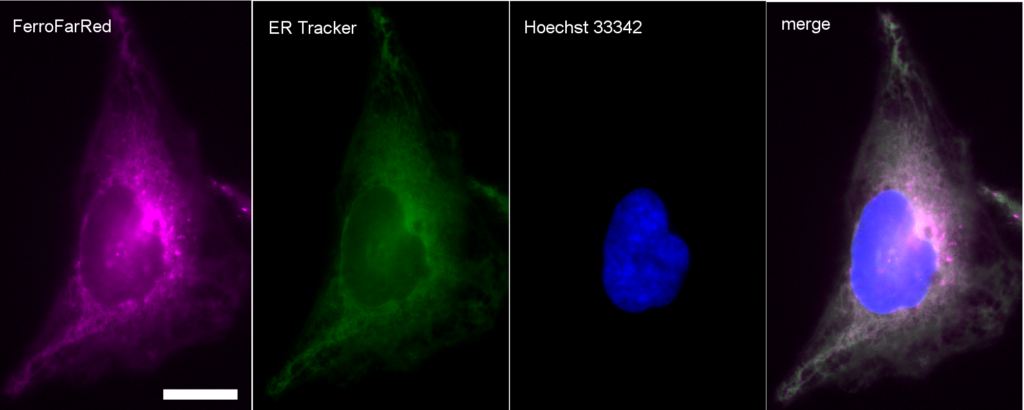
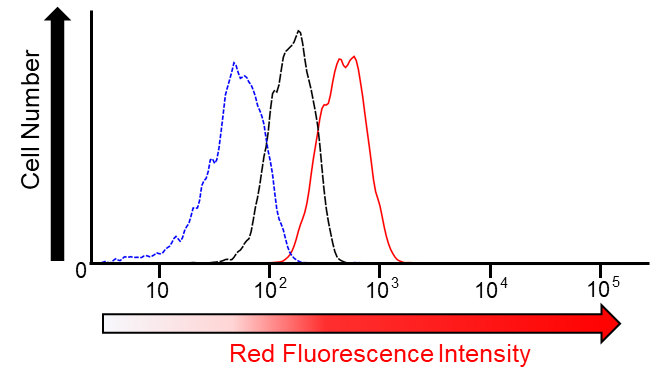
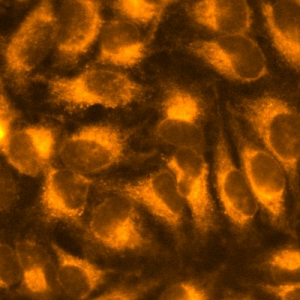
FerroFarRed (also known as SiRhoNox-1 or ER-SiRhoNox) is a fluorescent probe that specifically detects labile iron (II) ions (Fe2+). This probe is designed to selectively react with only Fe2+ separately from other metal ions and irreversibly turns into a far-red fluorescent substance. It mainly localizes inside endoplasmic reticulum (ER). It can also be used with a flow cytometer equipped with a red laser.
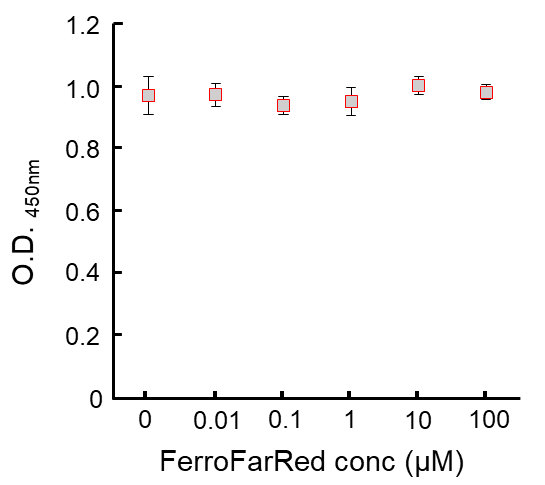
This probe does not have chelating effect.
Available through Merck KGaA (Darmstadt, Germany) as:
SCT037 BioTracker™ Far-red Labile Fe2⁺ Dye
![]()






 Contact Us
Contact Us
