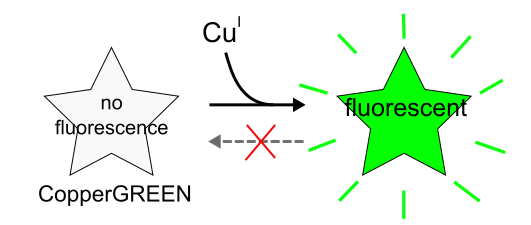
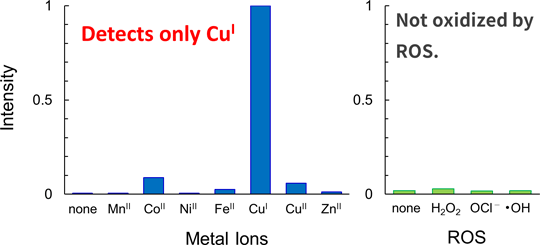
CopperGREEN™
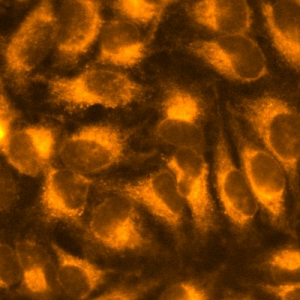
An example live-cell Imaging of copper
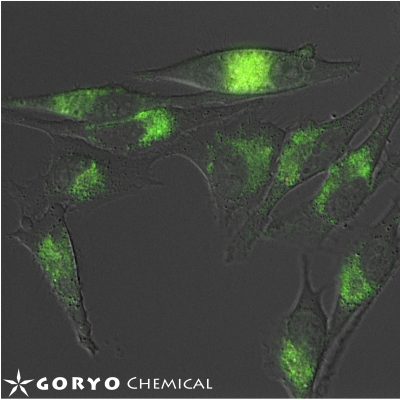
An example live-cell imaging using CopperGreen
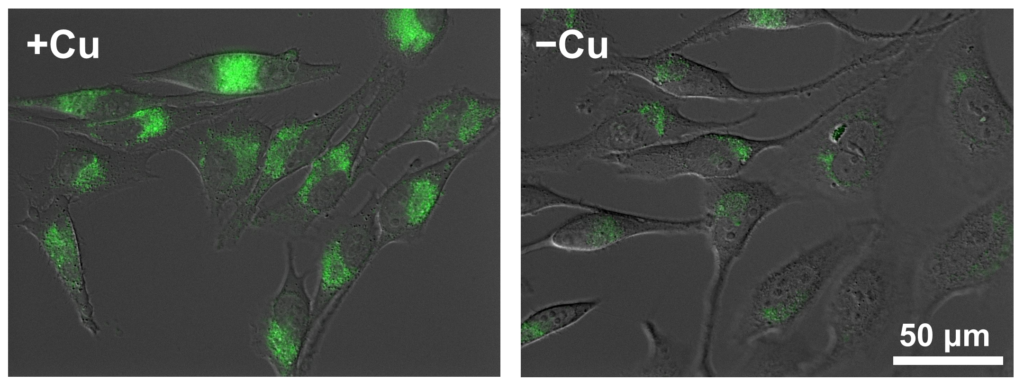
Fluorescence image of CopperGREEN (green) was overlaid to DIC image.
HeLa cells cultured in a culture medium (DMEM + 8% FBS, penicillin and streptomycin) supplemented with 200 µM CuCl2 were rinsed with PBS containing 200 µL EDTA for 2 times to remove extracellular copper ions. Then cells were reacted with 5 μM CopperGREEN at 37℃ 5% CO2 for 3 hours. For the fluorescence observation, the medium was exchanged with HBSS to reduce the background.
※ To reduce nonspecific staining, add 10 mM NH4Cl or 100 nM bafilomycin A1 to the medium 30 minutes before the staining and during the staining. This treatment inhibit acidification of lysosomes and reduces nonspecific oxidization of CopperGREEN in acidic environments. (10 mM NH4Cl was used in the above example.)
![]()






 Contact Us
Contact Us
