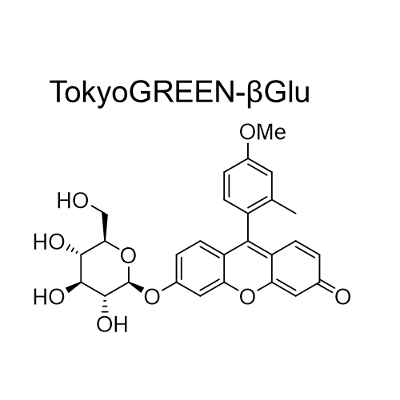
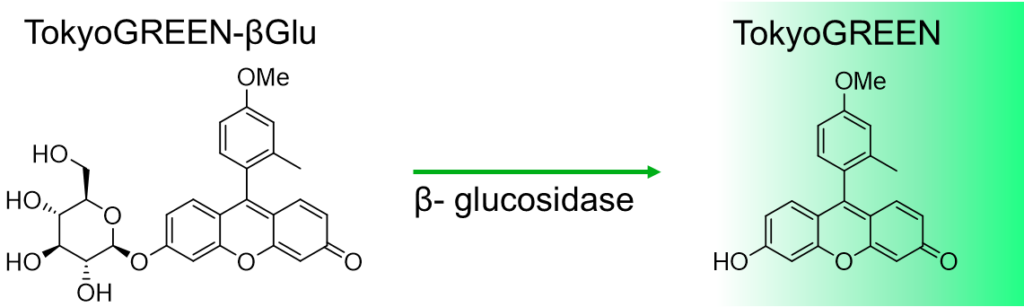
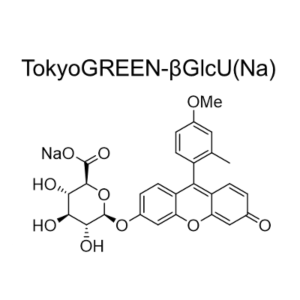
TokyoGreen–βGlu is fluorescent substrate [9- (4′-methoxy-2′-methylphenyl) -6- (β-D-glucopylanosyloxy) -xanthen-3-one] for β-glucosidase. Almost non-fluorescent TokyoGreen–βGlu is catalyzed by β-glucosidase, and generates bright fluorescent TokyoGreen. It is a cell permeable reagent and can detect intracellular glucosidase. TokyoGreen is also a cell permeable fluorophore, therefore fluorescence is observed both from the cells and extracellular fluid. Thus it is suitable for enzyme assay and for screening using plate reader.
Available through Merck KGaA (Darmstadt, Germany )as:
SCT026 BioTracker™ 510 Green β-Glu Dye
![]()




 Contact Us
Contact Us
