- HFP and AFP detects selectively highly reactive oxygen species (hROS) such as hydroxyl radical (・OH), peroxynitrite (ONOO-) and hypochlorite (OCl-), whereas they do not react with other reactive oxygen species (O-・2, H2O2, 1O2, NO, etc.).
- Quantitative analysis of H2O2 is available by adding HRP.
- Detecting specifically hypochlorite (OCl-) by the combination use of HPF and APF.
- HPF and APF is not autoxidized at all, which enables us to easily get more reliable data than by other reagents.
- Live fluorescence imaging of cells is available.
ROSFluor™ Series
Hydroxyphenyl Fluorescein (HPF) /Aminophenyl Fluorescein (APF)
[Reactive oxigen species (ROS) detecting probe]
Products
| Code No. | Product Name | Size | Merck CAT No. |
Merck ( Millipore / Sigma Aldrich ) Product Name |
|---|---|---|---|---|
| SK3001-02 | Hydroxyphenyl Fluorescein (HPF) | 1 mg | ||
| SK3002-02 | Aminophenyl Fluorescein (APF) | 1 mg |
-
Product Information
Hydroxyphenyl Fluorescein (HPF) /Aminophenyl Fluorescein (APF)
Product InformationPrint
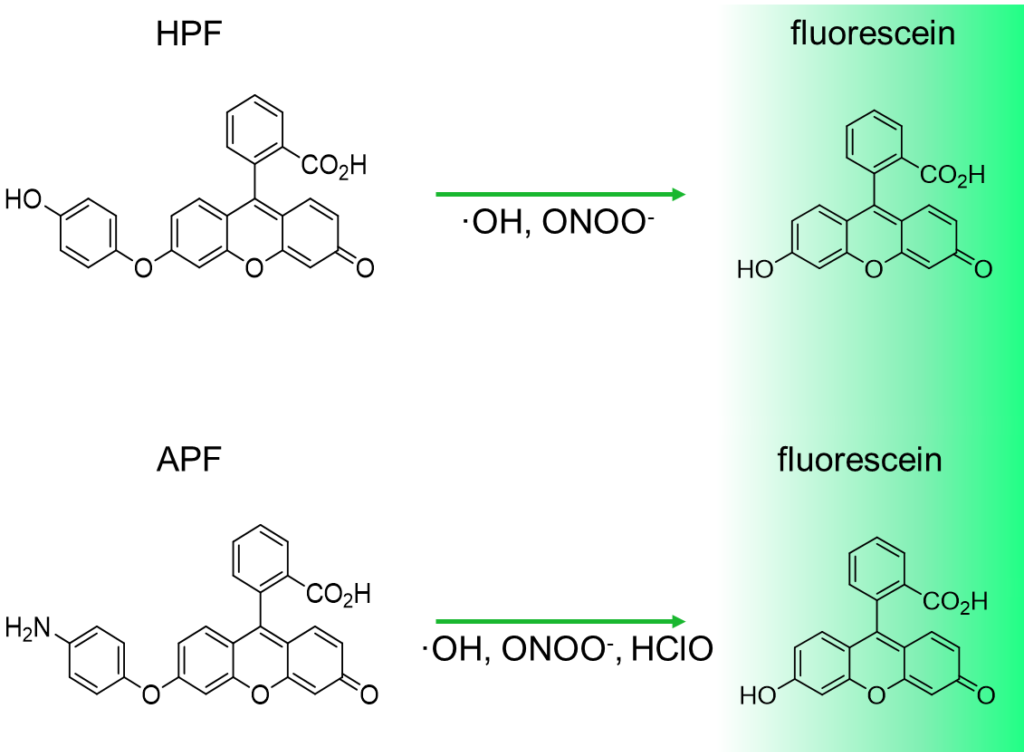
Reaction mechanism of HPF and APF
APF and HPF is almost non-fluorescent in neutral solutions. When they react with highly reactive oxygen species (hROS), they generate fluorescein that shows strong fluorescence (maximum excitation wavelength: 490 nm, maximum emission wavelength: 515 nm), and thus, increase in the fluorescence intensity is observed.
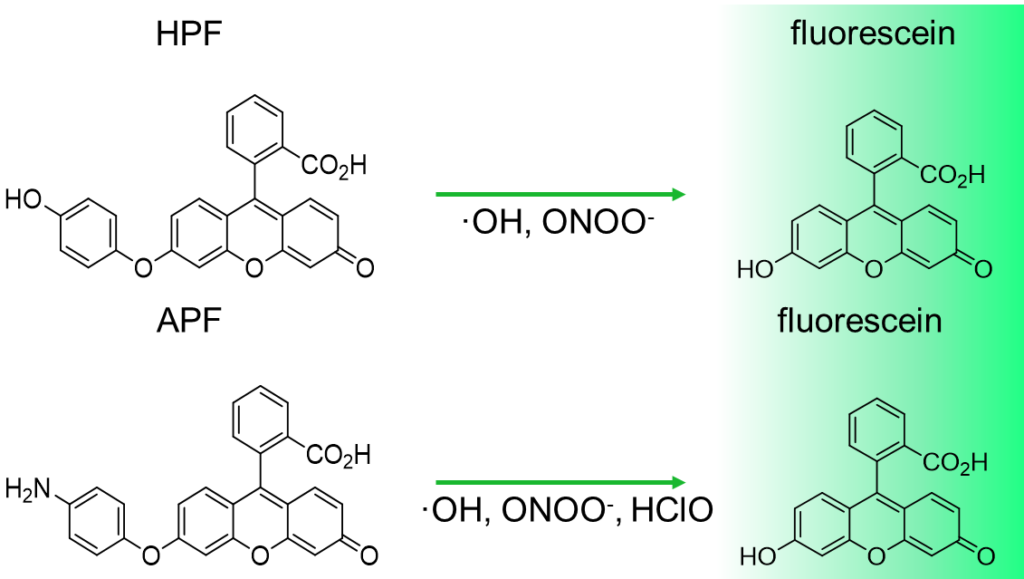
Reactivity and specificity of HPF, APF and DCFH
- Each fluorescent reagent (final concentration: 10 mM, added 0.1 % DMF as co-solvent) was reacted with various reactive oxygen species in phosphate buffer (0.1 M, pH 7.4).
- Fluorescent intensity of HPF, APF and DCFH was measured at 515, 515, 520 nm by the excitation of 490, 490, 500 nm, respectively.
Procedure
1 mg of HPF / APF is dissolve in N,N -dimethylformamide(DMF) 0.47 mL. Their densities are 5 mmol/L, each. Dilute the reagent 500~5000 times (10~1 μmol/L) by phosphate buffer (0.1 mol/L, pH 7.4) before use. Adding the diluted reagent to the sample and incubate, then measure the fluorescence of wave length: 515 nm with excitation of wave length: 490 nm.
-
Light-induced autoxidation of APF and HPF in cells
Hydroxyphenyl Fluorescein (HPF) /Aminophenyl Fluorescein (APF)
Light-induced autoxidation of APF and HPF in cellsPrint
Light-induced autoxidation of APF and HPF in cells
Adding fluorescent probe
- HeLa cells of 4×105 cell/mL were seeded to 3.5-cm dish and cultured overnight.
- 10 μM of APF, HPF, or DCFH-DA were added respectively, and incubated for 30 minutes at 37 ℃ in the dark.
Measuring fluorescence
- Observed by a confocal scanning microscope with excitation of 488 nm, and observing fluorescence of over 515 nm (before).
- Irradiated laser with the wavelength of 488 nm to the cells for 120 seconds, then observed again (after).
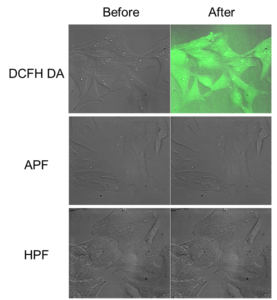
Autooxidation of DCFH-DA was observed after laser irradiation. In contrast, autooxidations of HPF and APF were not detected. Green fluorescence images and gray DIC images were overlayed.
-
A timecourse of ferroptosis observed by fluorescence live cell imaging using FerroOrange and ROSFluor series
Hydroxyphenyl Fluorescein (HPF) /Aminophenyl Fluorescein (APF)
A timecourse of ferroptosis observed by fluorescence live cell imaging using FerroOrange and ROSFluor seriesPrint
A timecourse of ferroptosis observed by fluorescence live cell imaging using FerroOrange and ROSFluor series
Ferroptosis is a cell death which depends on intracellular labile Fe2+. Its mechanism is known as a distinct from that of apoptosis or necrosis. Excess amount of intracellular labile Fe2+ induces generation of reactive oxigen species (ROS) and lipid peroxidation, those lead to cell death. It has also been revealed that ferroptosis is induced in some neurodegerative diseases, and some tumor cells are ferroptosis resistant.
Here, we tried to detect labile Fe2+ that induces ferroptosis, as well as ROS, by using activatable fluorescent probes of FerroOrange, APF, OxiORANGEand HYDROP, in the timecourse of ferroptosis.
Visualization of Fe2+ and ROS in the process of ferroptosis
Erastin was applied to HT-1080 cells to induce ferroptosis, and intracellular labile Fe2+ and ROS were imaged after 3, 6, 9 hours after the application, using the fluorescent probes. FerroOrange (1 μM), APF (5 μM), HYDROP (1 μM), and OxiORANGE (1 μM) were applied 30 minutes before each of the observation timepoint. Fluorescence signal of FerroOrange which indicates labile Fe2+ was maximum at 3 hours after the induction, whereas fluorescence of other ROS probes: APF which detects hydroxyradical (·OH), hypochlorous acid (HClO), and peroxinitrite (ONOO–), OxiORANGE which detects hydroxyradical (·OH), hypochlorous acid (HClO), and HYDROP which specifically detects hydrogen peroxide H2O2 were maximum at 6 hours after the induction. These results indicate that in ferroptosis, ROS increase after the increase in labile Fe2+.
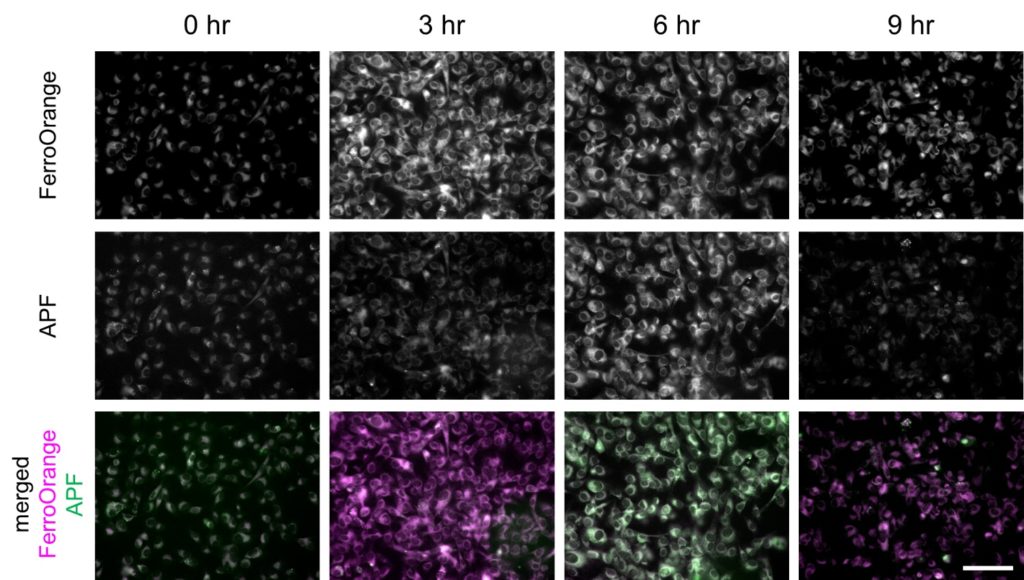
Fluorescence intensity changes of FerroOrange and APF in ferroptosis-induced HT-1080 cells
Fluorescence intensity of FerroOrange and APF in erastin-applied HT-1080 cells (upper and middle rows) were shown. Images were overlayed as pseudocolor images (bottom). Magenta indicates fluorescence of FerroOrange, green indicates that of APF. Bar indicates 100 µm.
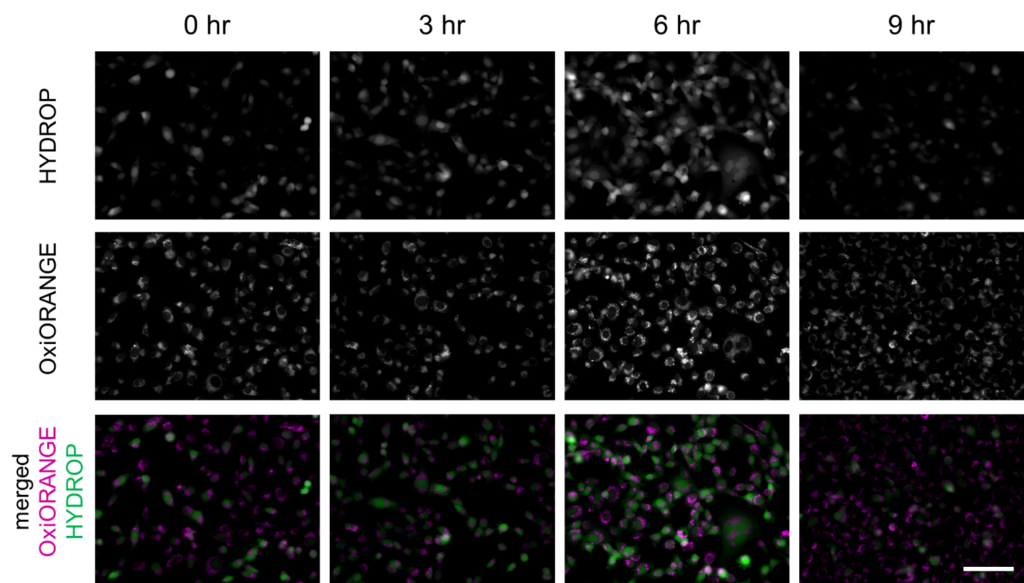
Fluorescence intensity changes of OxiORANGE and HYDROP in ferroptosis-induced HT-1080 cells
Fluorescence intensity of OxiOrange and HYDROP in erastin-applied HT-1080 cells (upper and middle rows) were shown. Images were overlayed as pseudocolor images (bottom). Magenta indicates fluorescence of OxiOrange, green indicates that of HYDROP. Bar indicates 100 µm.
Experiment procedure
- HT-1080 cells of 2 × 105 were seeded on glass bottom dishes and cultured until the cells attached to the bottom.
- Erastin (final concentration of 30 μM) was added to the media and cultured for2.5, 5.5, 8.5 hours at 37℃, 5% CO2.
- Each probes were added to the media and cultured for 30 minutes.
- The cells were rinsed twice with HBSS and observed by fluorescence microscopy.
※In different cell culture conditions, the concentrations of the reagents and incubation time might be varied. In this experiment, preliminary tests were needed to optimize those conditions.
※If the cell adhesion to the dishes was weak, we recommend to use poly-L-lysine coated dished.
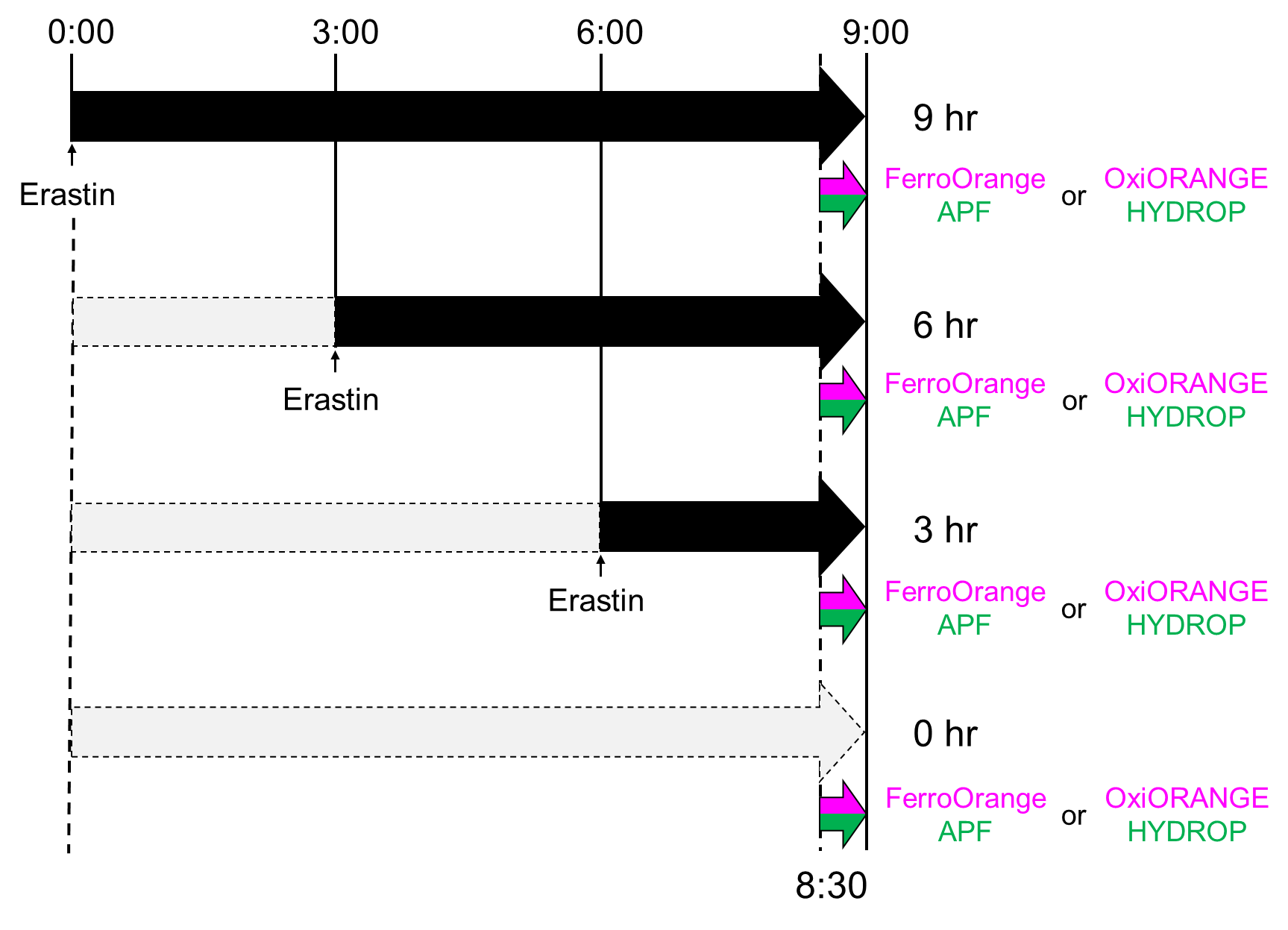
Actual timecourse of the experiment. Actually, erastin was added in different timepoints and addition of fluorescent probes and observations were performed simultaneously. Since these probes reacts with each target and fluoresces irreversibly, timecourse of ferroptosis was observed by the above procedure.
FAQ
-
Q What is the difference between APF and HPF?A
APF is more sensitive than HPF. HPF detects hydroxy radicals and peroxynitrates (・OH, ONOO–), wheras APF also detects hypochlorous acid (OCl–) in addition to them.
-
Q Are APF and HPF are cell permeable?A
Yes both of them are cell permeable. Therefore both can be used to detect intracellular reactive oxygen species.
-
Q Can APF/HPF be used in vivo?A
HPF can be used for in vivo imaging in rat cerebral cortex. Please refer the following paper.
Y. Wang, S. Yamamoto, A. Miyakawa, T. Sakurai, K. Ibaraki, S. Terakawa (2010)
Neurosurgery. 67: 112-118 DOI: 10.1227/01.NEU.0000370055.99998.6BHPF also can be used in frozen section and brain slice.
In some cases, APF could not be applied to the sample to which HPF can be applied.
-
Q Can I use DMSO for dissolving OxiORANGE, HYDROP, APF, HPF?A
We recommend to use DMF for these reagents because DMSO is known to be a quencher of one of the reactive oxygen species (ROS), hydroxy radicals. Even though you are intended to detect aother ROS, the ROS could be generated by the signal of hydroxy radicals and usage of DMSO could also decrease the target ROS. Therefore we recommend to use DMF instead of DMSO.
-
Q Tell me a selection guide of ROSFluor series.A
Product code Product name Exmax (nm) Emmax (nm) ・OH ONOO- HClO H2O2 O2-・ ROS detection in solutions ROS detection in cells GC3004-01 OxiORANGE 553 577 + - + - - + + GC3006-01 HySOx 553 574 - - + - - + + GC3007-01 HYDROP 492 518 - - - + - – + GC3008-01 HYDROP-EX※ 492 518 - - - + - + – SK3001-01 HPF 490 515 + + - - - + + SK3002-01 APF 490 515 + + + - - + + SK3003-01 NiSPY-3 490 515 - + - - - + + ※HYDROP-EX is suitable for detection of extracellular H2O2 since it has low cell permeability.
-
Q My question is not in this FAQ list.....A
Please also refer the frequently asked questions on fluorescent probes






 Contact Us
Contact Us
