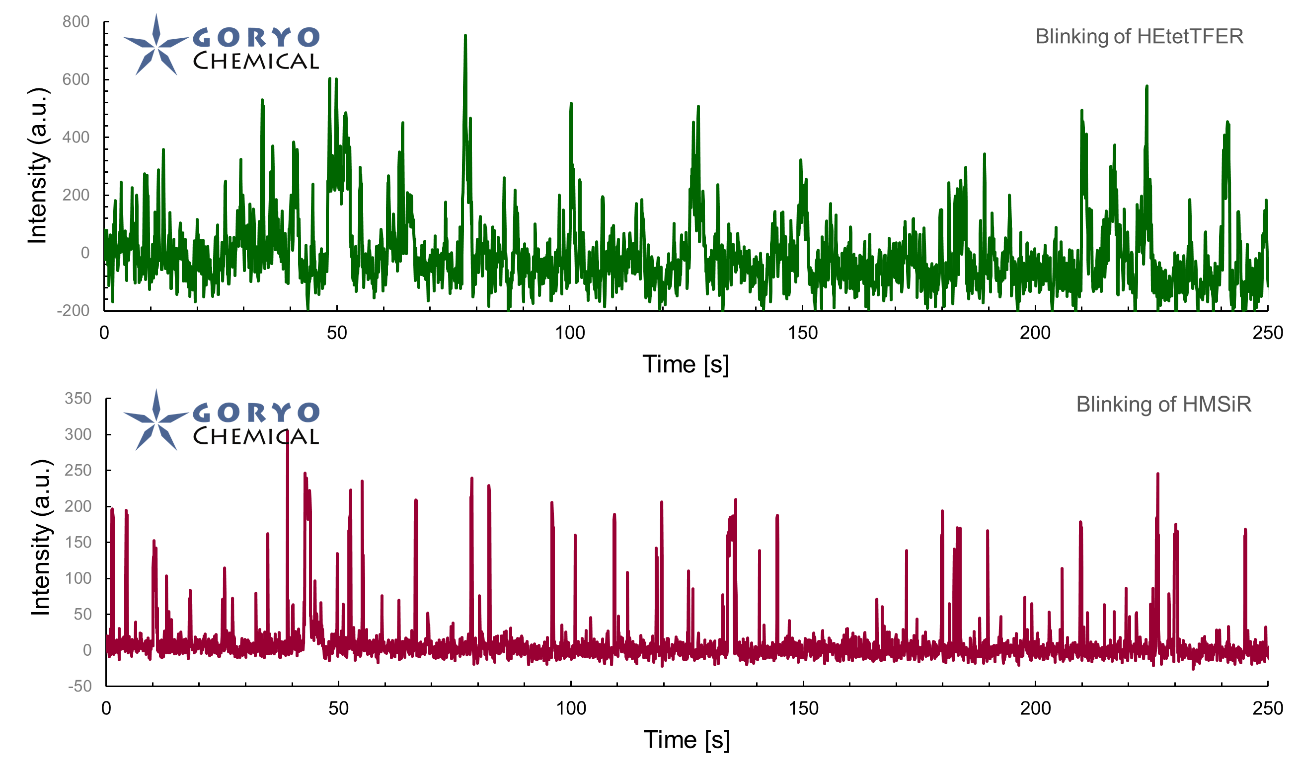
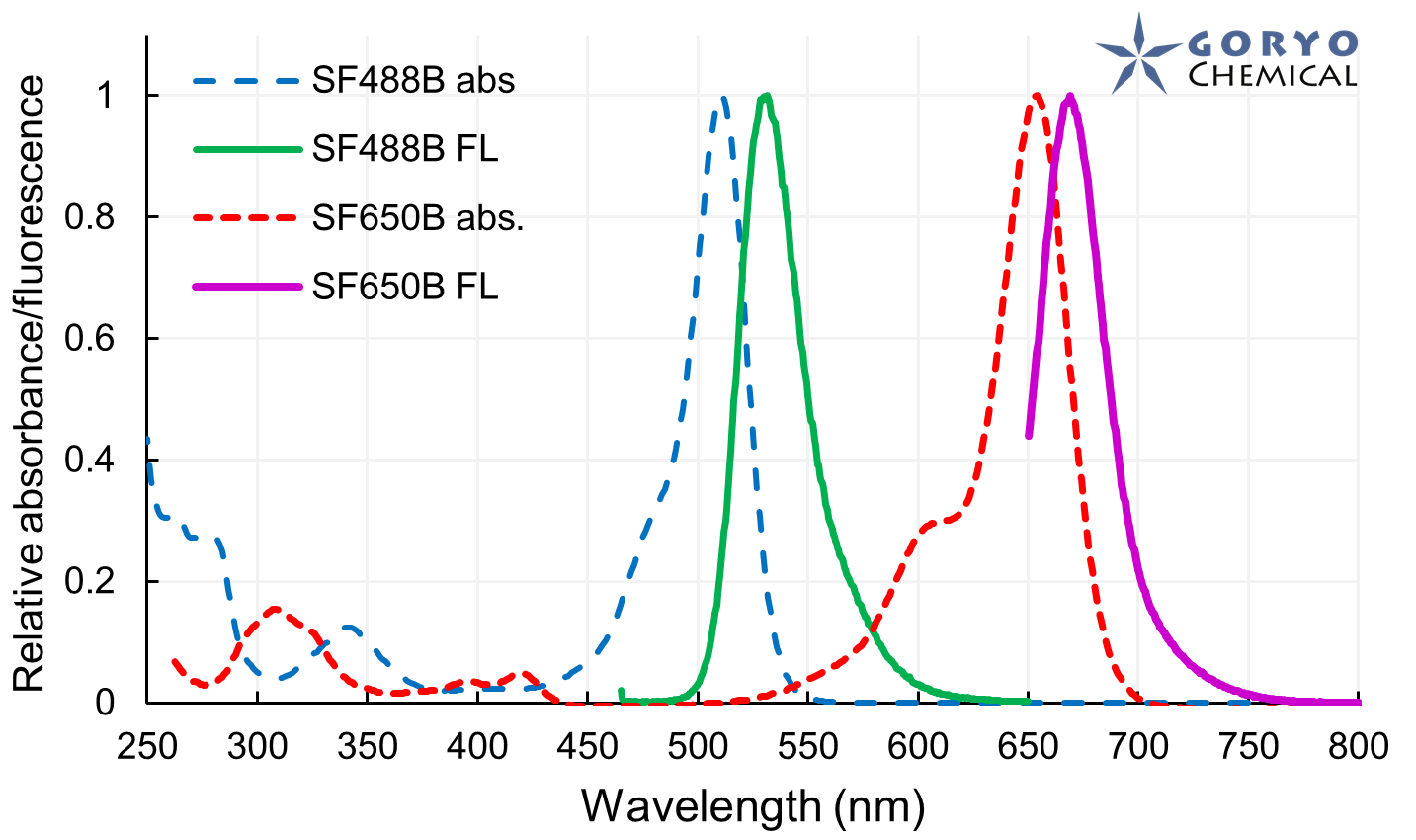
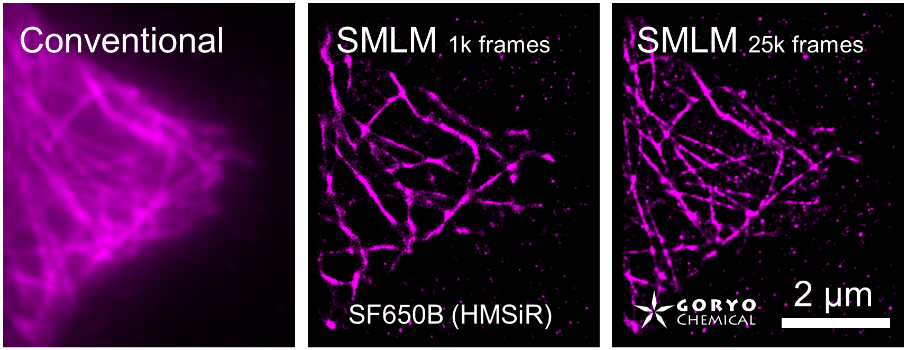
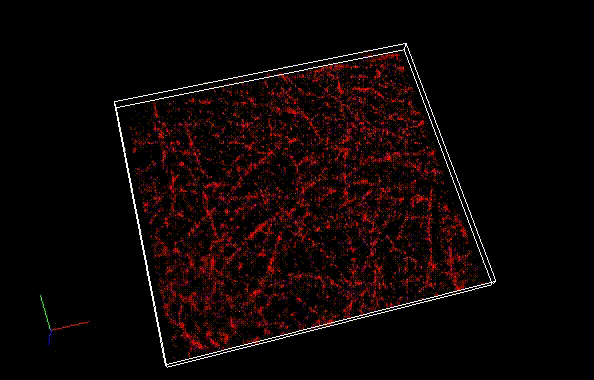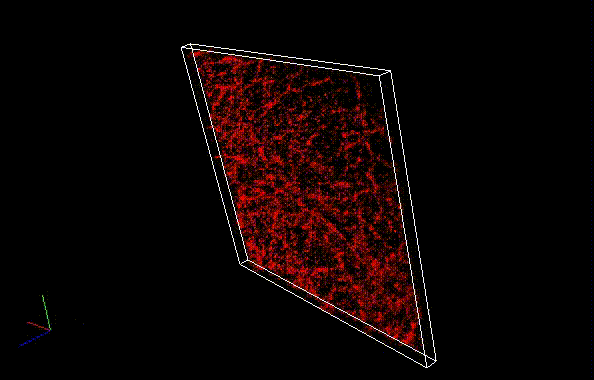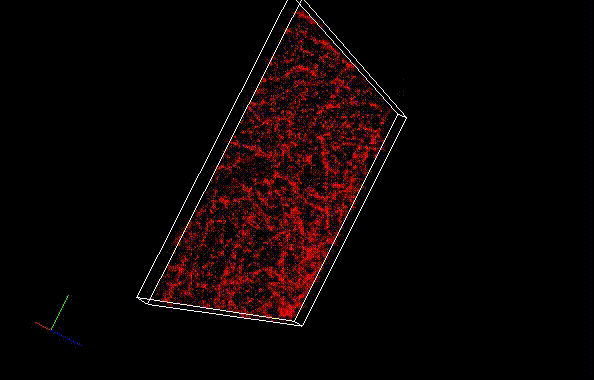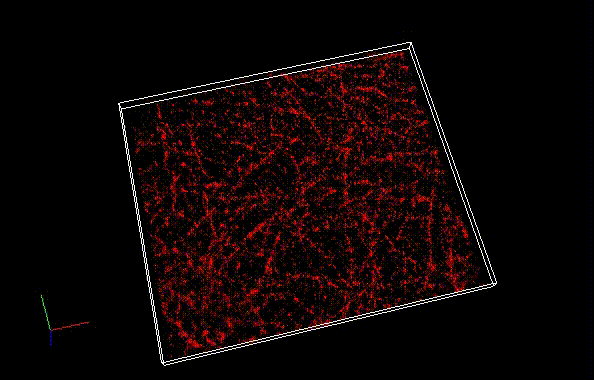
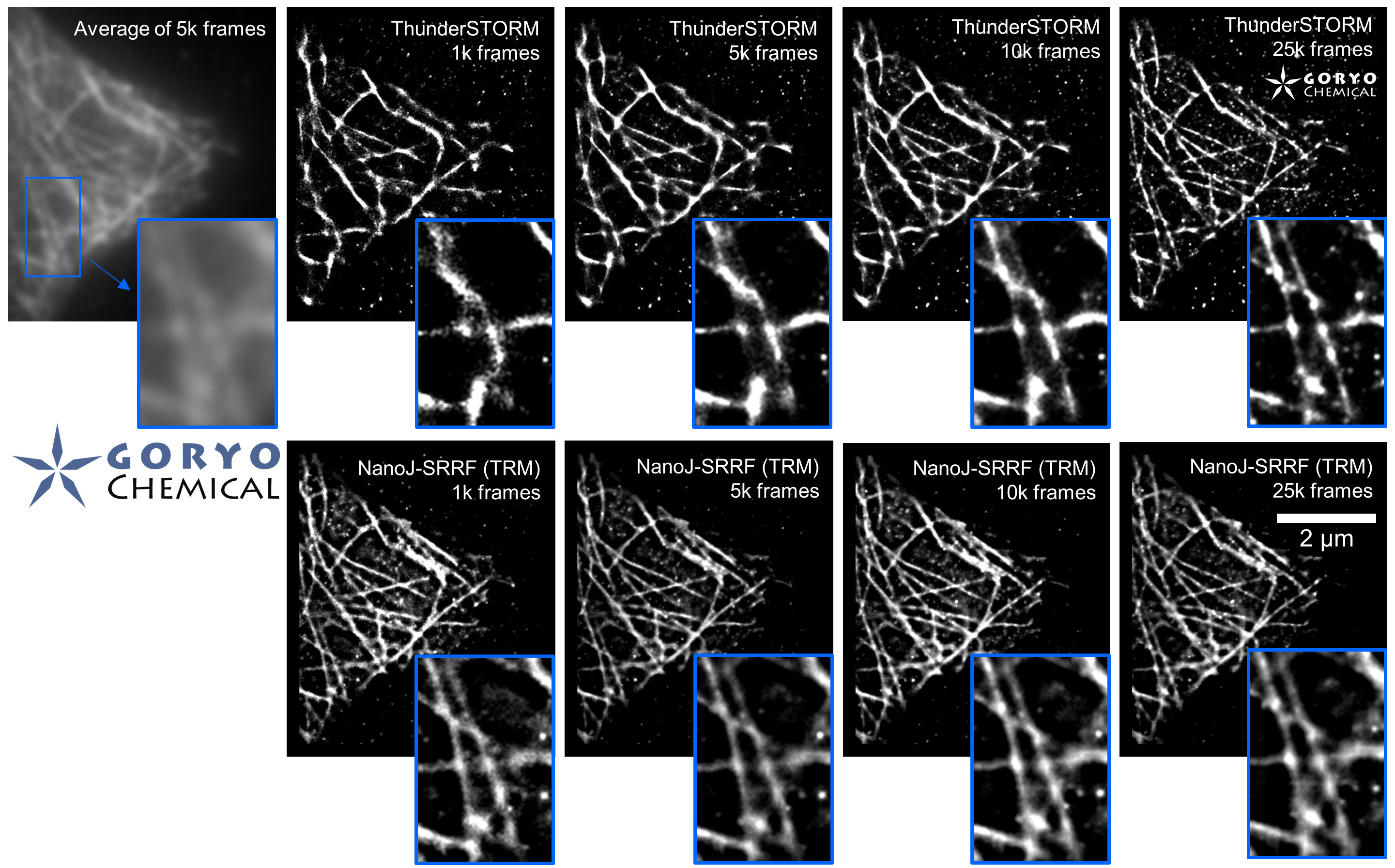
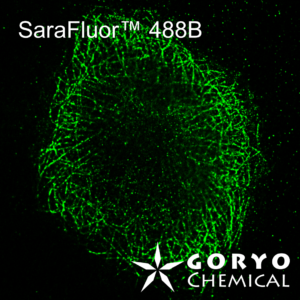
The theoretical limit of the resolution of fluorescence microscopy imaging is about half of the light wavelength (200-400 nm). A number of imaging methods called “superresolution fluorescence microscopy” exceeding this limitation have been developed. This fluorophore series has been developed for single molecule localization microscopy (SMLM), one of the superresolution microscopy methods. The SaraFluor B series is a fluorescent probe that exhibits spontaneous blinking under physiological conditions. This ability allows for super-resolution imaging using STORM, PALM and other similar microscopy systems under physiological conditions. It does not require the addition of thiols, deoxidizing agents or exposure to strong laser radiation to induce blinking as with general fluorescent probes. These products are secondary antibodies for immunofluorescent staining.
* SaraFluor is named after an Ainu language word “sara” which means “become visible” or “spacious and bright wetland”. Probes which exhibit spontaneous blinking are marked with letter B, first letter of the word “blinking”.
** These products are the same as those sold under the name of HMSiR labeling Goat IgG (whole) anti-mouse / rat / rabbit IgG H&L.







 Contact Us
Contact Us
