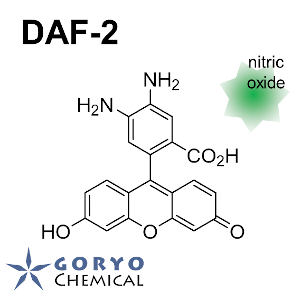
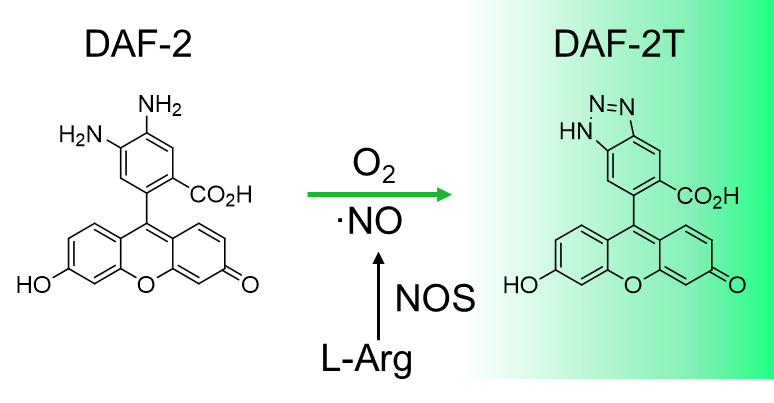
Diaminofluorescein-2(DAF-2)is a fluorescent probe to detect nitrogen oxide (NO). It shows almost no fluorescence but strongly fluoresces upon reaction with NO. It is cell impermeable reagent and suitable for the detection of extracellular NO.
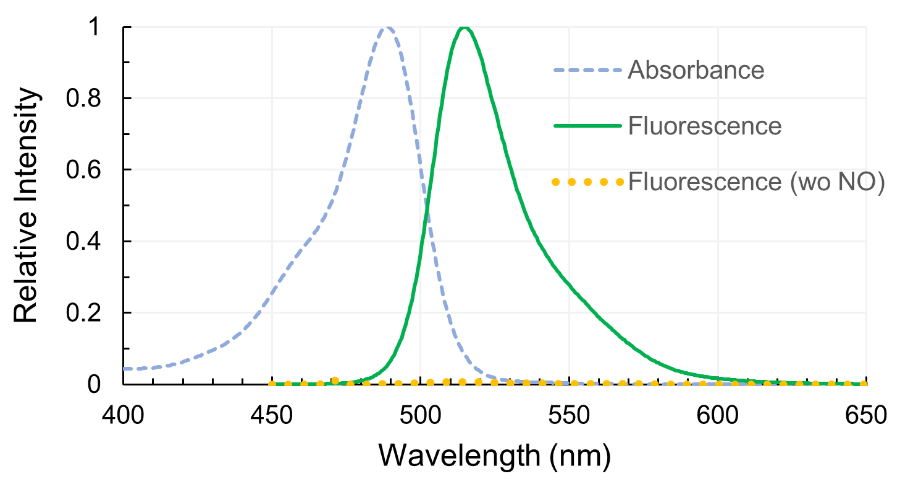
Yes. Excitation max at 495 nm and fluorescence max at 515 nm are the wavelength of each peaks. It can be measured at the wavelengths of slightly differs (Anal. Chem. 1998, 70, 2450). It is often observed that excitation at the wavelength shorter than the peak and detection at the wavelength longer than the peak lower the background and make better detection efficiency. Also refer the manual of the plate reader since it depends on the properties of the detector.







 Contact Us
Contact Us
