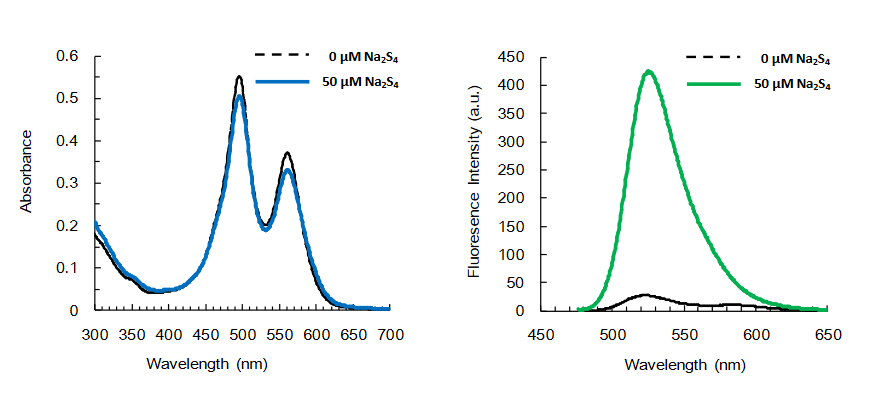
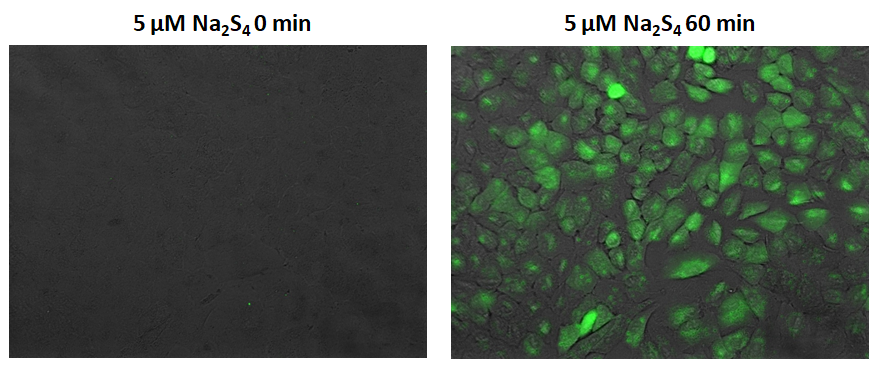
Intracellularly abundant sulfane sulfur species including persulfide (R-S-SH), polysulfide (R-S-Sn-S-R) and polysulfide (H2Sn) play important roles to maintain intracellular reducing environments. SSip-1 DA is a FRET-based fluorescence probe to specifically detect the intracellular sulfane sulfur species. It shows only week fluorescence without sulfane sulfur, and reversibly fluoresces in response to micromolar concentrations of sulfane sulfur. Its reaction is highly specific and has minimal to no reactivity with H2S, cysteine residues, and sulfur oxides. SSip-1 DA, a cell permeable diacetylated form of SSip-1 is hydrolyzed by intracellular esterases to generate SSip-1 which retains within the cells. Thus it is suitable to monitor intracellular concentration of sulfane sulfur via live-cell imaging.
Available through Merck KGaA (Darmstadt, Germany) as:
SCT211 BioTracker Green Sulfane Sulfur Live Cell Dye
![]()








 Contact Us
Contact Us
