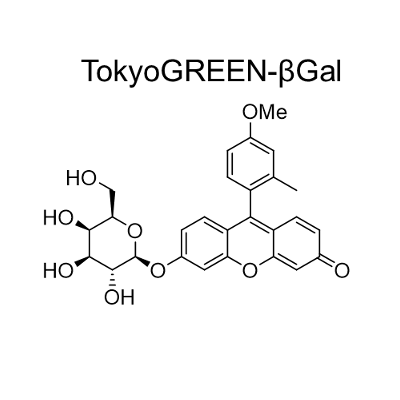
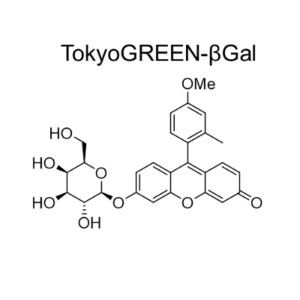
TokyoGreen®-βGal is cell permeable fluorescent substrate (9-(4′-methoxy-2′-methylphenyl)- 6-(β-D-galactopylanosyloxy) -xanthen-3-one) for detecting β-galactosidase. Cell lysis or fixation is not required since TokyoGreen-βGal is easily incorporated into cells. TokyoGreen-βGal is applicable to the experiments of gene expression test, gene targeting and cell cloning utilizing lacZ gene marker.
GlycoFluor™ Series
Products
| Code No. | Product Name | Size | Merck CAT No. |
Merck ( Millipore / Sigma Aldrich ) Product Name |
|---|---|---|---|---|
| SK4001-01 | TokyoGreen®–βGal | 1 mg |
-
Product Information
TokyoGreen®–βGal
Product InformationPrint
Principle of the measurement
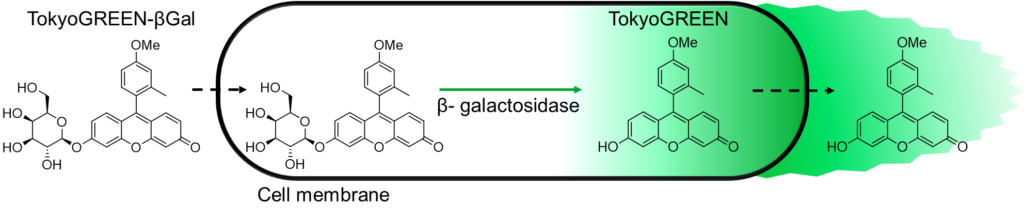
Non-fluorescent TokyoGreen–βGal is taken into the cell, is hydrolyzed by the β-galactosidase, and generates bright fluorescent TokyoGreen. TokyoGreen is also permeable through the cell membrane, so that generated TokyoGreen diffuses uniformly in the culture medium and that the whole medium makes green fluorescence (510 nm) when it is irradiated by the 490 nm excitation light.
Contents
TokyoGreen–βGal 1mg (5 mM in DMSO 0.4mL)
C27H26O9MW:494.49
-
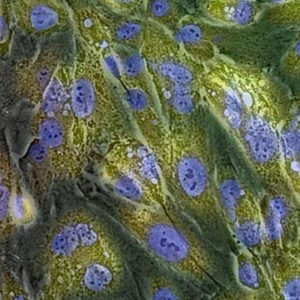
Detecting β-galactosidase activity of fixed cells using TokyoGreen™-βGal
TokyoGreen®–βGal
Detecting β-galactosidase activity of fixed cells using TokyoGreen™-βGalPrint
Detecting β-galactosidase activity of fixed cells using TokyoGreen™-βGal
TokyoGreen-βGal is a fluorescent substrate for β-galactosidase. This product shows 400 times stronger fluorescence upon reaction with β-galactosidase. This property can be applied to monitor the expression of LacZ reporter gene or for detection of other β-galactosidase activities. This product is highly cell permeable before and after the reaction with β-galactosidase, then after the reaction with β-galactosidase-positive cells, fluorescence intensities of whole medium increases. Therefore this product is most suitable for screening in a plate-reader format. Since the application protocol is simple, it is suitable for screening assays.
This product is applicable both for living cells and fixed cells, here we introduce an application example for fixed cells.
Reaction procedure
- Fix HEK 293 cells cultured on glass bottom dish for 10 to 30 minutes with 3% of paraformaldehyde.
* Fixing time more than 30 minutes is not recommended as the fluorescent intensity might decrease. - Wash the fixed cells gently with HBSS once.
- Dilute TokyoGREEN®–βGal by HBSS or appropriate observation buffer to adjust final concentration of 2.5 µM.
- Add the staining solution to the washed HEK 293 cell, and incubate for 30 minutes at 37℃.
- Observe by standard method
Please do not wash the cells once you stained with TokyoGREEN®–βGal which is membrane permeable and fluoresce also in the culture medium.
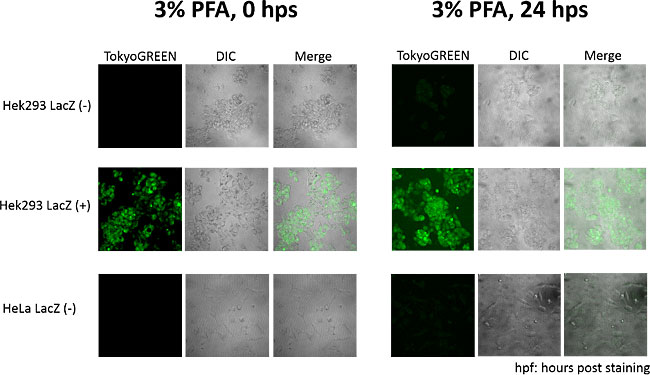
Fluorescence images taken by confocal laser scanning microscopy at 0 hour and 24 hours after fixation and staining.
(Left) Fluorescence images at 0 hour after fixation and staining: Strong fluorescence was observed on LacZ expressing cells. Fluorescence is hardly observed on no LacZ expressing cells.(Right) Fluorescence images at 0 hour after fixation and staining: Clear difference between LacZ expressing cells and no LacZ expressing cells still can be observed.
* Imaging using conventional fluorescence microscope instead of confocal laser microscope.
- Fix HEK 293 cells cultured on glass bottom dish for 10 to 30 minutes with 3% of paraformaldehyde.
FAQ
-
Q Give me a selection guide of the β-galactosidase probes.A
We have three β-galactosidase probes, GlycoGREEN-βGal, GlycoGREEN-βGal, and TokyoGreen-βGal . For cell imaging, we recommend GlycoGREEN-βGal, for plate-reader assays or for enzyme kinetics measurements, we recommend TokyoGreen-βGal as a first choice.
Sensitivity of the probe isGlycoGREEN-βGal ~ TokyoGreen-βGal > GlycoYELLOW-βGal.
For cell imaging, we recommend GlycoGREEN-βGal because its cell retention is relatively better, however, generated fluorophore also leaks from the cells and can be incorporated into other cells, users should carefully check the result when they observe heterogeneous cells. -
Q Can I use for the fixed samples?A
You may use a similar procedure to X-gal staining for fixed cell samples, because GlycoYELLOW-βGal is also a substrate for β-galactosidase (lacZ reporter)
We confirmed that fluorescence of GlycoYELLOW-βGal observed in live cells does not diminish by the normal process of fixation. Please refer information in the product page for details.
Strong fixation conditions may alter the intracellular localization or may decrease the fluorescence. Please test the fixation condition in your cells and conditions.
-
Q Can I detect senescence?A
Yes, using GlycoYELLOW-βGal, you can detect enhancement of β-galactosidase activity caused by senescence (SA-βGal).
If physiological β-Gal activity in lysosomes is detected with GlycoYELLOW-βGal, then you may use bafilomycin A1 for alkalization of lysosomes and inhibit physiological β-Gal activity. You may not need to use alkalization reagent if you do not detect physiological β-Gal.
-
Q My question is not in this FAQ list.....A
Please also refer the frequently asked questions on fluorescent probes




 Contact Us
Contact Us
