Detection of phagocytosis using AcidiFluor ORANGE-Zymosan A with a flow cytometer
Summary
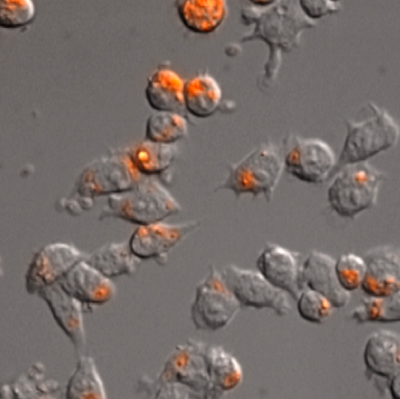
Zymosan A is a compound prepared from yeast cell walls. It is often used to induce inflammatory responses, such as phagocytosis. AcidiFluor ORANGE-Zymosan A is Zymosan A labeled with AcidiFluor ORANGE, an acidic pH indicator. Its fluorescence intensity is highest in an acidic environment, such as phagosomes. AcidiFluor ORANGE-Zymosan A is a useful probe to measure phagocytosis, since the increase in fluorescence intensity only occurs after phagocytosis. Here we present an example data to evaluate phagocytosis of mouse macrophage-derived RAW 264.3 cells using AcidiFluor ORANGE-Zymosan A, and comparison with other products.
Phagocytotic activity of RAW264.7 cells measured using a flow cytometer
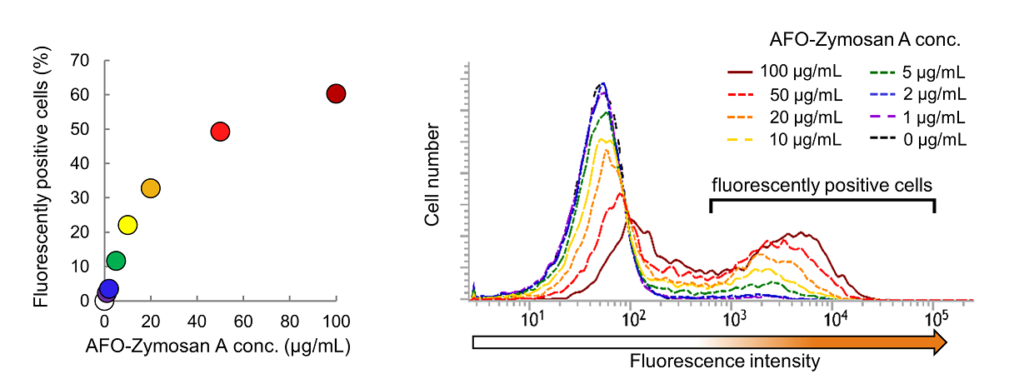
Fig. 1. Phagocytotic activity of RAW264.7 cells upon the concentration of AcidiFluor ORANGE-Zymosan A (AFO-Zymosan A). Each concentration of AcidiFluor ORANGE-Zymosan A was added to RAW264.7 cells, then incubated at 37°C, 5% CO2 for 1 hour. Cells were rinsed with PBS and analyzed. Left shows the summarized result, whereas the right shows the fluorescence intensity distribution in each condition. The number of fluorescently positive cells were determined as the number of cells which fluorescence intensity is above 6 × 102. AcidiFluor ORANGE was excited with a 488-nm laser, and the fluorescence intensity was measured with a 586/42 nm filter for phycoerythrin.
Protocol
- Seed 7.5×104 cells of RAW264.7 in a 12-well plate and incubate overnight.
- Suspend AcidiFluor ORANGE-Zymosan A in cell culture medium at 50 µg/mL.
- Sonicate the suspension for 2 minutes to disperse AcidiFluor ORANGE-Zymosan A, and put on ice every 2 minutes. Repeat this step 5 times.
- Remove the culture medium from the well plate and add the AcidiFluor ORANGE-Zymosan A suspension to the cells.
- Incubate cells at 37°C, 5% CO2 for 1 – 2 hours to induce phagocytosis.
- Remove the AcidiFluor ORANGE-Zymosan A suspension and rinse with PBS twice.
- Detach cells with trypsin and neutralize with the medium containing serum.
- Centrifuge the cell suspension, discard the supernatant, and resuspend the cells in PBS.
- Filter the cells through a cell strainer (40-µm nylon mesh) to remove debris.
- Analyze the sample using flow cytometry.
|
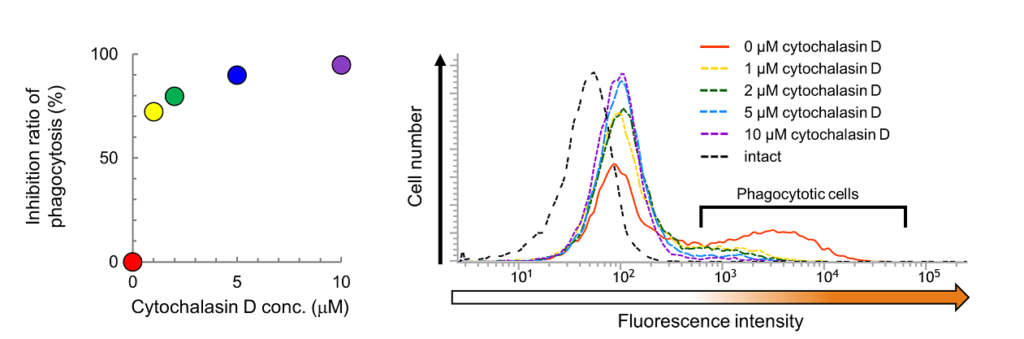
Fig. 2 Effect of cytochalasin D on phagocytosis examined using AcidiFluor ORANGE-Zymosan A. After pretreatment of cytochalasin D (phagocytosis inhibitor) at various concentrations (0 µM: red, 1 µM: yellow, 2 µM: green, 5 µM: blue, and 10 µM: purple), AcidiFluor ORANGE-Zymosan A of 50 µg/mL was added to RAW264.7 cells and incubated at 37°C, 5% CO2 for 1 hour. The cells rinsed with PBS were analyzed using a flow cytometer.Left indicates the summarized result, while the right indicates the distribution of fluorescence intensity in each condition. The number of fluorescence-positive cells indicated in the right plot are determined by the number of cells which fluorescence intensity is above 6 × 102. The inhibition ratio of phagocytosis (ri) was calculated as follows:
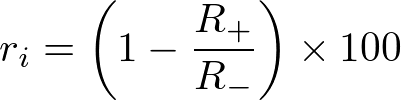
where R+ represents the percentage of phagocytotic cells treated with cytochalasin D, and R- represents the percentage of phagocytotic cells without the addition of cytochalasin D. AcidiFluor ORANGE was excited with a 488-nm laser, and the fluorescence intensity was measured with 586/42 nm filter for Phycoerythrin.
These data indicate that the fluorescence intensity profile well reflects the efficiency of phagocytosis by macrophages.
Comparison with other products
Bright fluorescence of AcidiFluor ORANGE-Zymosan A, in addition to its high signal-to-noise ratio, enables easier discrimination of phagocytotic cells from non-phagocytotic cells. It gives higher signal intensity compared with other pH indicating fluorophores, such as pHrodo Red and pHrodo Green.
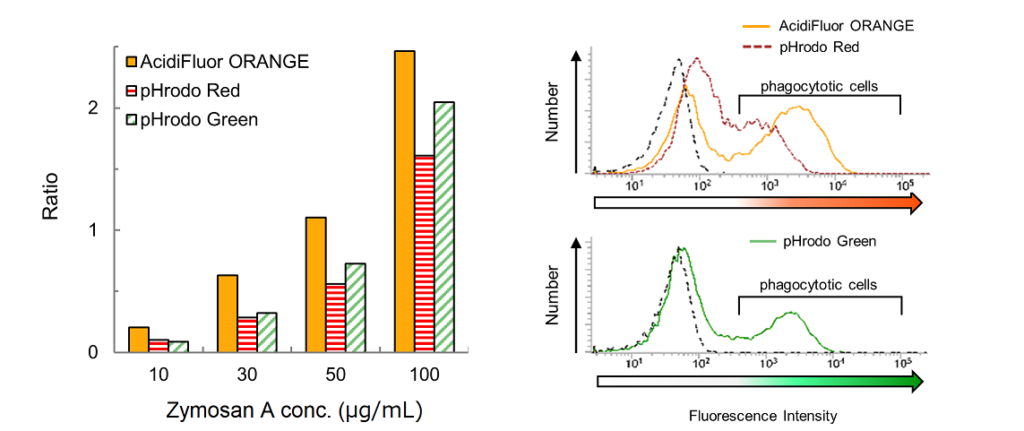
Fig. 3 Comparison with pHrodo Red-Zymosan A and pHrodo Green-Zymosan A. Various concentrations (10, 30, 50, 100 µg/mL) of AcidiFluor ORANGE-Zymosan A, pHrodo Red-Zymosan A, and pHrodo Green-Zymosan A were added to RAW264.7 cells and incubated at 37°C, 5% CO2 for 1 hour. The cells rinsed with PBS were analyzed using flow cytometry. Left indicates the ratio of phagocytotic cells in each concentration of Zymosan A labeled with each fluorophores. Right indicates a representative distribution of fluorescence intensity measured at the concentration of 50 µg/mL. Fluorescence intensity above 3 × 102 units are determined as fluorescence positive cells. AcidiFluor ORANGE, pHrodo Red, and pHrodo Green were excited with a 488-nm laser. The fluorescence intensities of AcidiFluor ORANGE and pHrodo Red were measured with a 586/42 nm filter for Phycoerythrin, while that of pHrodo Green was measured with 537/32 nm filter for FITC. The negative control (a non-treated sample) is indicated as black broken lines.
As shown in Fig. 3, the ratio between the phagocytotic cells and the non-phagocytotic cells was the highest for AcidiFluor ORANGE-Zymosan A (left) at every concentration. In addition, AcidiFluor ORANGE-Zymosan A could better separate the phagocytotic cells from non-phagocytotic cells in fluorescence intensity distribution when compared with pHrodo Red-Zymosan A (right).








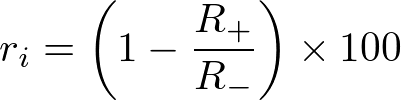

 Contact Us
Contact Us
