About QuicGSH3.0
QuicGSH3.0 is a fluorescent probe to quantify intracellular reduced gulutathione concentration, by using intramolecular FRET mechanism. (FRET = fluorescence resonance energy transfer)
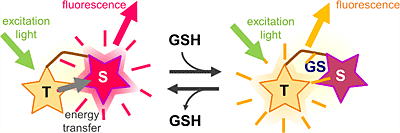 QuicGSH3.0 is a combined molecule of orange fluorophore T and deep-red fluorophore S. When the concentration of GSH is low, green excitation light excite the fluorophore T, and the excited energy is transfered to the second fluorophore S to emit deep-red fluorescence. When the concentration of GSH is high and GSH binds to the fluorophore S, the energy-transfer is inhibited and therefore it emits orange fluorescence.
QuicGSH3.0 is a combined molecule of orange fluorophore T and deep-red fluorophore S. When the concentration of GSH is low, green excitation light excite the fluorophore T, and the excited energy is transfered to the second fluorophore S to emit deep-red fluorescence. When the concentration of GSH is high and GSH binds to the fluorophore S, the energy-transfer is inhibited and therefore it emits orange fluorescence.
Regardress of the probe concentration, the ratio of the orange fluorescence intensity and the deep-red fluorescence intensity is determined only be the GSH concentation. Therefore by measureing the fluorescence intensity of the two wavelength range, users can determine intracellular GSH concentation.
Since the dissociation constant of QuicGSH3.0 to GSH (Kd) is 3.0 mM, it is suitable to determine the millimolar range of intracellular GSH concentartions. The fluorescence intensity ratio rapidly changes upon the GSH concentration changes, users can trace the real-time intracellular GSH concentration changes.
Properties of QuicGSH3.0
| Product Name |
Target |
Reactivity |
Absmax (nm) |
FLmax (nm) |
| QuicGSH3.0 |
GSH |
reversible |
558 |
582, 625 |
Spectral changes depending on the GSH concentration
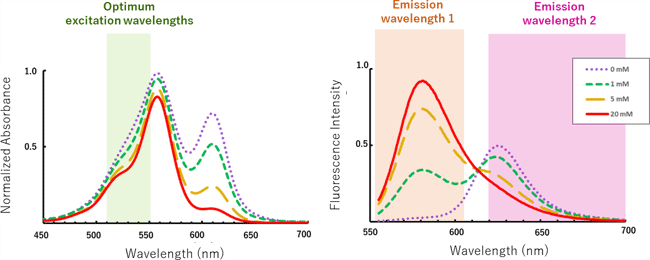
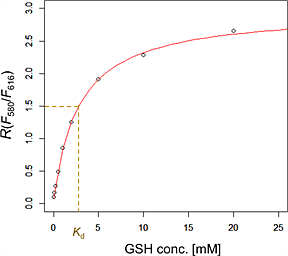
The emission maximum (λem) is 625 nm in the absence of GSH, whereas it shifts to 582 nm upon addition of GSH. Fluorescent intensity ratio (R = F580/F616) increases when GSH is increased.
Quickness and reversibility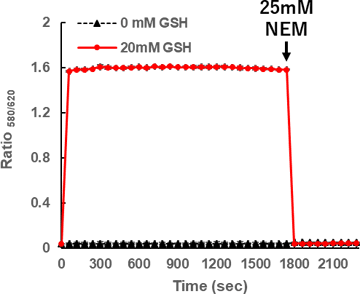
QuicGSH3.0 rapidly reacts with GSH to shift its λem from 625 to 580 nm. λem also returns quickly to 625 nm when GSH is quenched.
2 µM QuicGSH3.0 (in 0.2 M phosphate buffer, pH 7.4, containing 5% DMSO as cosolvent) was incubated with GSH at 27ºC. At 1800 seconds later, a thiol scavenger, N-ethylmaleimide (NEM) was added. Fluorescence intensities were recorded every 60 seconds and the ratio of the fluorescence intensities at 580 nm and 620 nm were plotted (λex: 550 nm, λem: 580 nm and 620 nm, n=3 ± S.D.).
![]()






 QuicGSH3.0 is a combined molecule of orange fluorophore T and deep-red fluorophore S. When the concentration of GSH is low, green excitation light excite the fluorophore T, and the excited energy is transfered to the second fluorophore S to emit deep-red fluorescence. When the concentration of GSH is high and GSH binds to the fluorophore S, the energy-transfer is inhibited and therefore it emits orange fluorescence.
QuicGSH3.0 is a combined molecule of orange fluorophore T and deep-red fluorophore S. When the concentration of GSH is low, green excitation light excite the fluorophore T, and the excited energy is transfered to the second fluorophore S to emit deep-red fluorescence. When the concentration of GSH is high and GSH binds to the fluorophore S, the energy-transfer is inhibited and therefore it emits orange fluorescence.


 Contact Us
Contact Us
