H.Jiang , R. K. Muir, R. L. Gonciarz, A. B. Olshen, I. Yeh, B. C. Hann, N. Zhao, Y. H. Wang , S. C. Behr, J. E. Korkola, M. J. Evans, E. A. Collisson, A. R. Renslo (2022)
J. Exp. Med. 219: e20210739. DOI: 10.1084/jem.20210739
D. Y. Kang, N. Sp, E. S. Jo, J. M. Lee. K. J. Jang (2021)
Cells 10: 2549-2562 DOI: 10.3390/cells10102549
Fumoto, E. Kinoshita,K. Ohta, K. Nakamura,T. Hirayama, H. Nagasawa, D. Hu, K. Okami, R. Kato, S. Shimokawa, N. Ohira, K. Nishimura, H. Miyamoto, T. Tanaka, S. Kawakami, K. Nishida (2020)
Pharmaceutics 12: 1070 DOI: 10.3390/pharmaceutics12111070
Z.Li, L. Jiang, S. H. Chew, T. Hirayama, Y. Sekido, S. Toyokuni (2019)
Redox Biol. 26: 101297 DOI: 10.1016/j.redox.2019.101297
D. Cui, M. Arima, T. Hirayama, E. Ikeda (2019)
Exp. Cell Res. 379 : 166-171 DOI: 10.1016/j.yexcr.2019.04.003.
K. Sato, L. Shi, F. Ito, Y. Ohara, Y. Motooka, H. Tanaka, M. Mizuno, M. Hori, T. Hirayama, H. Hibi, S. Toyokuni (2019)
J. Clin. Biochem. Nutr. 65 : 8-15 DOI : 10.3164/jcbn.18-91
T. Imai, S. Iwata, T. Hirayama, H. Nagasawa, S. Nakamura, M. Shimazawa H. Hara (2019)
Sci. Rep. 9: 6228-6243 DOI: 10.1038/s41598-019-42370-z
T. Hirayama, A. Miki, H. Nagasawa (2018)
Metallomics. in press DOI: 10.1039/c8mt00212f
K. Sakamoto, T. Suzuki, K. Takahashi, T. Koguchi, T. Hirayama, A. Mori, T. Nakahara, H. Nagasawa, K. Ishii (2018)
Exp. Eye Res. 171: 30-36 DOI: 10.1016/j.exer.2018.03.008
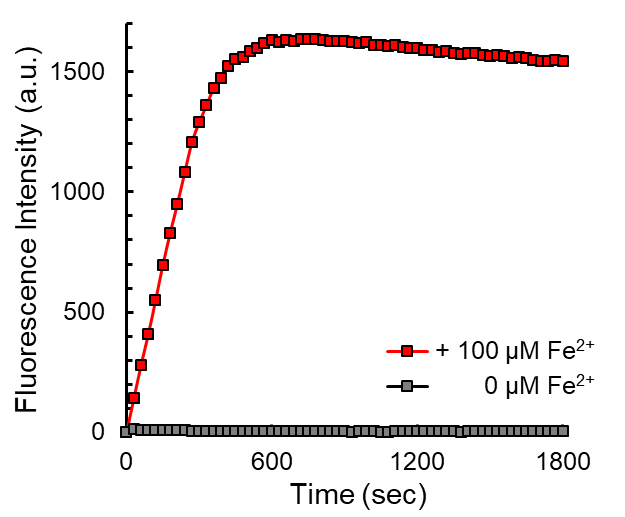
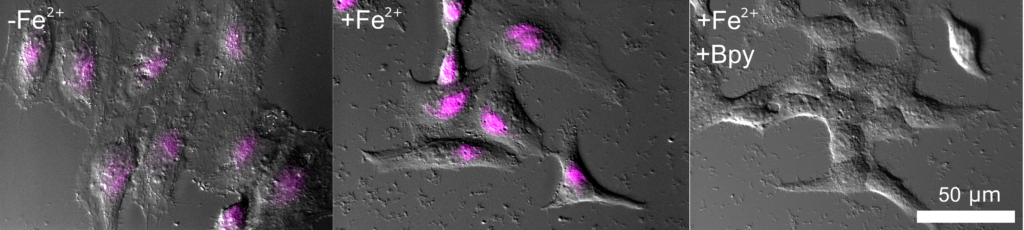
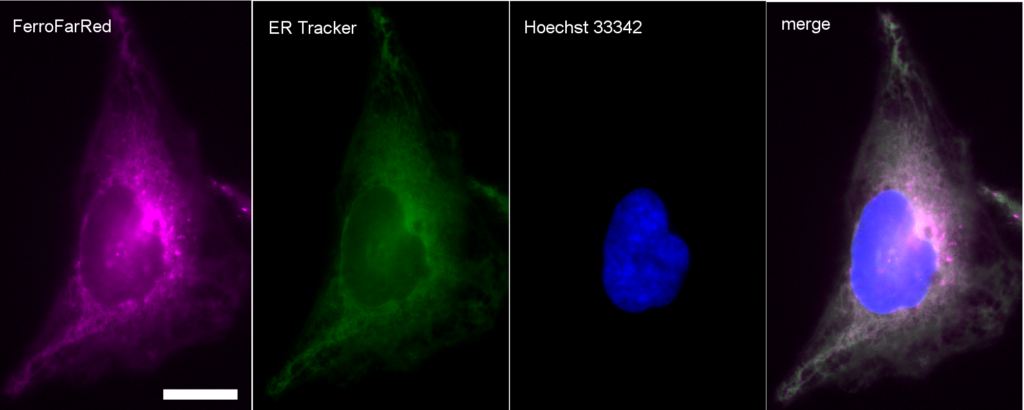
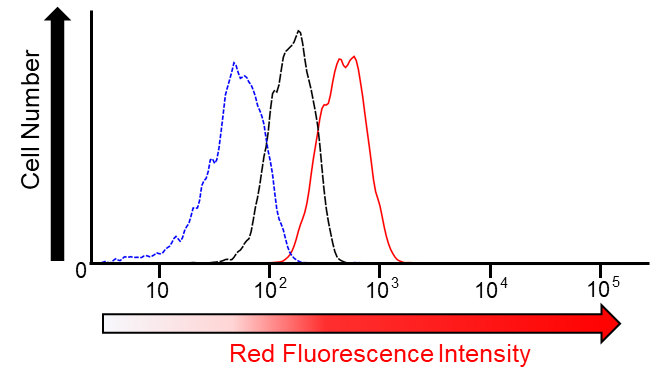
T. Hirayama, H. Tsuboi, M. Niwa, A. Miki, S. Kadota, Y. Ikeshita, K. Okuda, H. Nagasawa (2017)
Chem. Sci. 8: 4858-4866 DOI: 10.1039/c6sc05457a
![]()


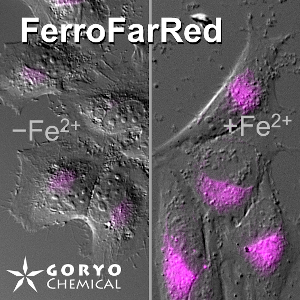



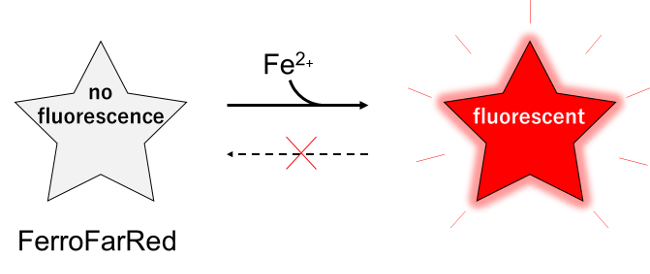
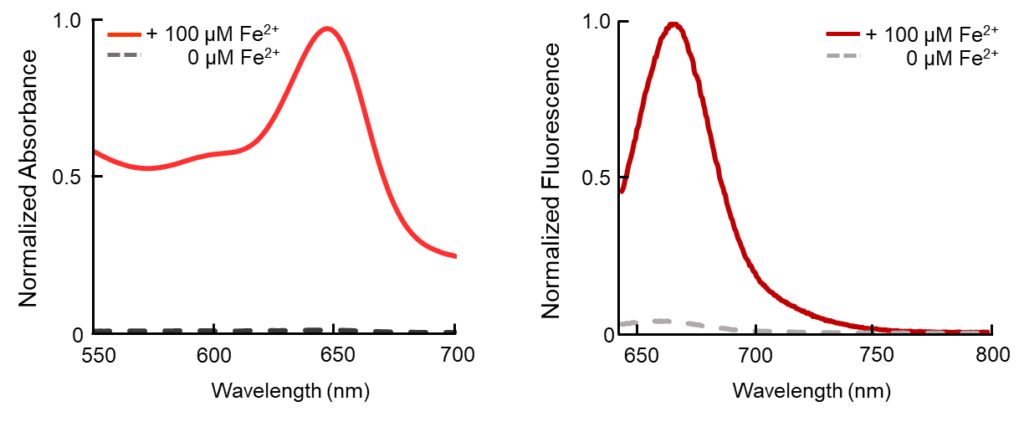
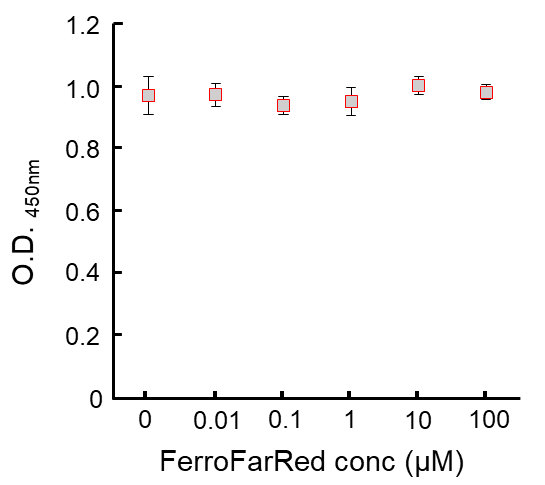

 Contact Us
Contact Us
