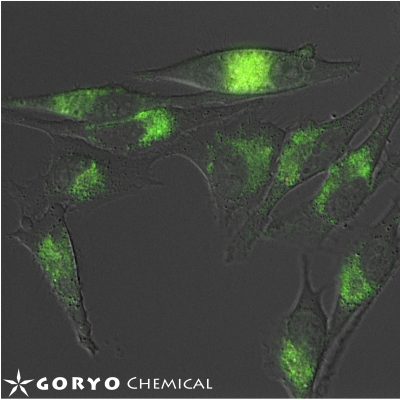
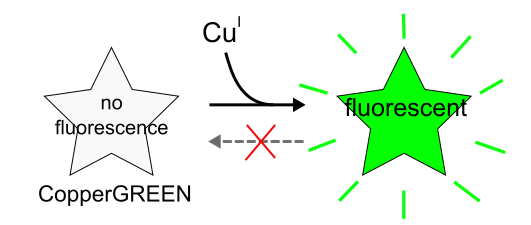
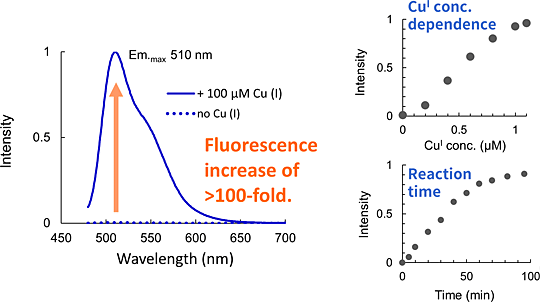
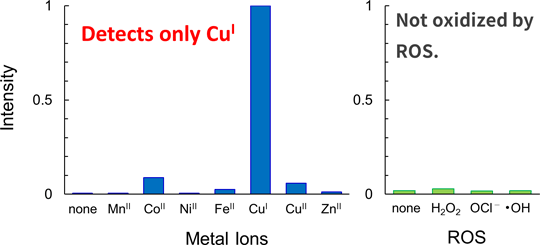
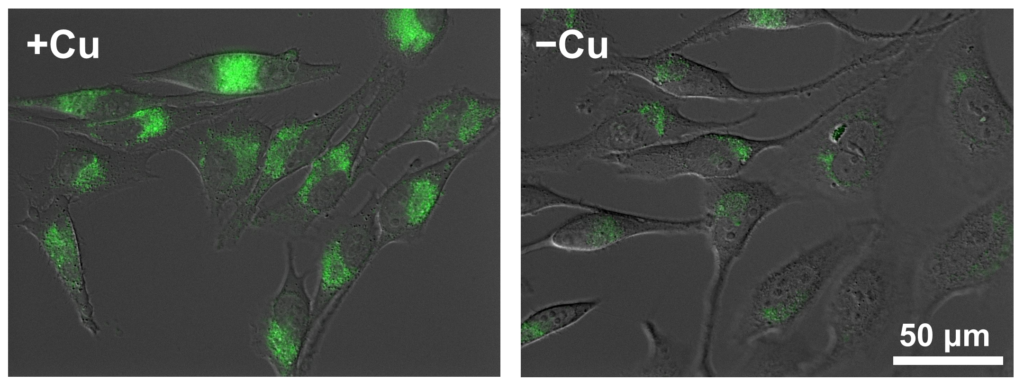
Copper is one of the trace metals essential for living organisms. Copper is an essential cofactor for enzyme activities of cytochrome c, superoxide dismutase, and tyrosinase. CopperGREEN is a fluorescent probe to detect Cu+, which is a dominant redox state of copper in an intracellular reducing environment.
Available through Merck KGaA (Darmstadt, Germany) as:
SCT041 BioTracker™ Green Copper Dye
![]()





 Contact Us
Contact Us
